Crystal structure of phosphoenolpyruvate carboxylase from Arabidopsis thaliana in complex with aspartate and citrate
Connell, M.B., Lee, M.J.Y., Plaxton, W.C., Jia, Z.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Phosphoenolpyruvate carboxylase 3 | 987 | Arabidopsis thaliana | Mutation(s): 0 Gene Names: PPC3, PEPC, PPC, At3g14940, K15M2.8 EC: 4.1.1.31 | 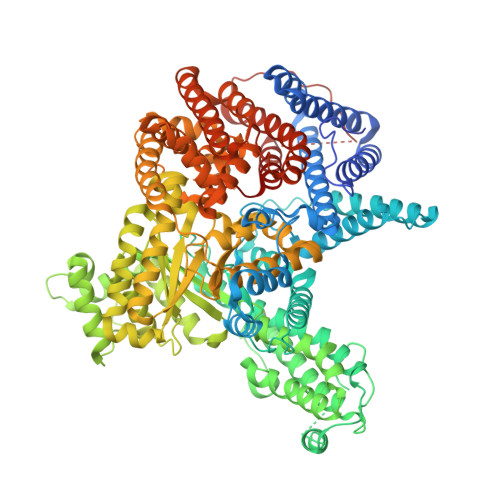 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q84VW9 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| FLC Download:Ideal Coordinates CCD File | D [auth A], F [auth B] | CITRATE ANION C6 H5 O7 KRKNYBCHXYNGOX-UHFFFAOYSA-K |  | ||
| ASP Download:Ideal Coordinates CCD File | C [auth A], E [auth B] | ASPARTIC ACID C4 H7 N O4 CKLJMWTZIZZHCS-REOHCLBHSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 264.26 | α = 90 |
| b = 268.13 | β = 90 |
| c = 77.93 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| Blu-Ice | data collection |
| XDS | data reduction |
| PHASER | phasing |
| XDS | data scaling |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Natural Sciences and Engineering Research Council (NSERC, Canada) | Canada | RGPIN203705-2013 |