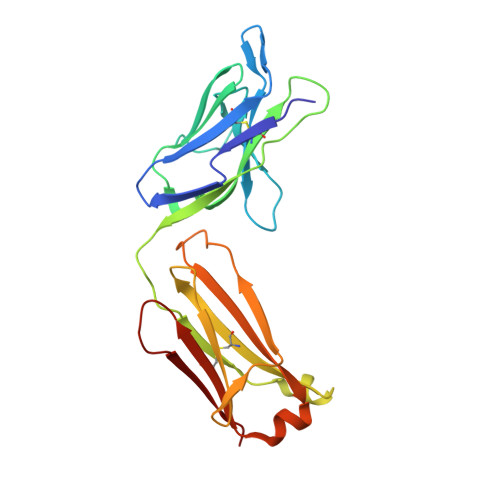
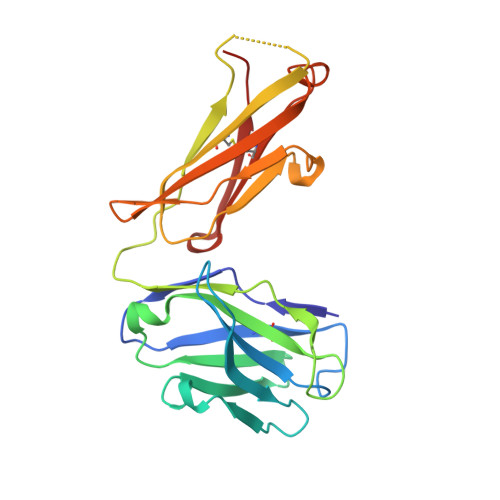
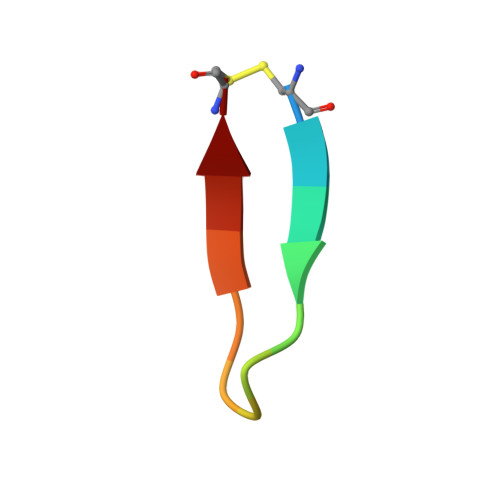
Generation and Characterization of Monoclonal Antibodies against a Cyclic Variant of Hepatitis C Virus E2 Epitope 412-422.
Sandomenico, A., Leonardi, A., Berisio, R., Sanguigno, L., Foca, G., Foca, A., Ruggiero, A., Doti, N., Muscariello, L., Barone, D., Farina, C., Owsianka, A., Vitagliano, L., Patel, A.H., Ruvo, M.(2016) J Virol 90: 3745-3759
- PubMed: 26819303
- DOI: https://doi.org/10.1128/JVI.02397-15
- Primary Citation Related Structures:
5EOC - PubMed Abstract:
The hepatitis C virus (HCV) E2 envelope glycoprotein is crucial for virus entry into hepatocytes. A conserved region of E2 encompassing amino acids 412 to 423 (epitope I) and containing Trp420, a residue critical for virus entry, is recognized by several broadly neutralizing antibodies. Peptides embodying this epitope I sequence adopt a β-hairpin conformation when bound to neutralizing monoclonal antibodies (MAbs) AP33 and HCV1. We therefore generated new mouse MAbs that were able to bind to a cyclic peptide containing E2 residues 412 to 422 (C-epitope I) but not to the linear counterpart. These MAbs bound to purified E2 with affinities of about 50 nM, but they were unable to neutralize virus infection. Structural analysis of the complex between C-epitope I and one of our MAbs (C2) showed that the Trp420 side chain is largely buried in the combining site and that the Asn417 side chain, which is glycosylated in E2 and solvent exposed in other complexes, is slightly buried upon C2 binding. Also, the orientation of the cyclic peptide in the antibody-combining site is rotated by 180° compared to the orientations of the other complexes. All these structural features, however, do not explain the lack of neutralization activity. This is instead ascribed to the high degree of selectivity of the new MAbs for the cyclic epitope and to their inability to interact with the epitope in more flexible and extended conformations, which recent data suggest play a role in the mechanisms of neutralization escape. Hepatitis C virus (HCV) remains a major health care burden, affecting almost 3% of the global population. The conserved epitope comprising residues 412 to 423 of the viral E2 glycoprotein is a valid vaccine candidate because antibodies recognizing this region exhibit potent neutralizing activity. This epitope adopts a β-hairpin conformation when bound to neutralizing MAbs. We explored the potential of cyclic peptides mimicking this structure to elicit anti-HCV antibodies. MAbs that specifically recognize a cyclic variant of the epitope bind to soluble E2 with a lower affinity than other blocking antibodies and do not neutralize virus. The structure of the complex between one such MAb and the cyclic epitope, together with new structural data showing the linear peptide bound to neutralizing MAbs in extended conformations, suggests that the epitope displays a conformational flexibility that contributes to neutralization escape. Such features can be of major importance for the design of epitope-based anti-HCV vaccines.
- Institute of Biostructures and Bioimaging, CNR and CIRPeB, University of Naples Federico II, Naples, Italy.
Organizational Affiliation: