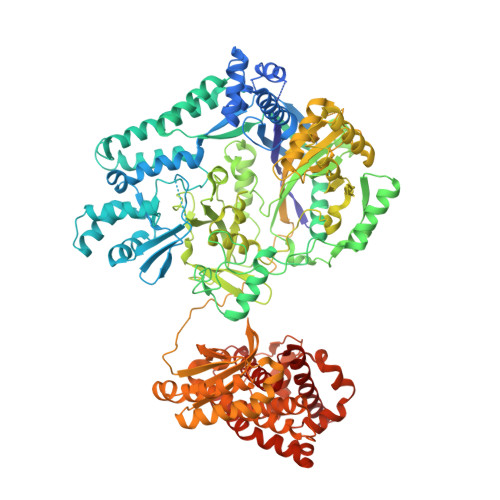
Structure and tRNA Specificity of MibB, a Lantibiotic Dehydratase from Actinobacteria Involved in NAI-107 Biosynthesis.
Ortega, M.A., Hao, Y., Walker, M.C., Donadio, S., Sosio, M., Nair, S.K., van der Donk, W.A.(2016) Cell Chem Biol 23: 370-380
- PubMed: 26877024 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.chembiol.2015.11.017
- Primary Citation Related Structures:
5EHK - PubMed Abstract:
Class I lantibiotic dehydratases dehydrate selected Ser/Thr residues of a precursor peptide. Recent studies demonstrated the requirement of glutamyl-tRNA(Glu) for Ser/Thr activation by one of these enzymes (NisB) from the Firmicute Lactococcus lactis. However, the generality of glutamyl-tRNA(Glu) usage and the tRNA specificity of lantibiotic dehydratases have not been established. Here we report the 2.7-Å resolution crystal structure, along with the glutamyl-tRNA(Glu) utilization of MibB, a lantibiotic dehydratase from the Actinobacterium Microbispora sp. 107891 involved in the biosynthesis of the clinical candidate NAI-107. Biochemical assays revealed nucleotides A73 and U72 within the tRNA(Glu) acceptor stem to be important for MibB glutamyl-tRNA(Glu) usage. Using this knowledge, an expression system for the production of NAI-107 analogs in Escherichia coli was developed, overcoming the inability of MibB to utilize E. coli tRNA(Glu). Our work provides evidence for a common tRNA(Glu)-dependent dehydration mechanism, paving the way for the characterization of lantibiotics from various phyla.
- Department of Biochemistry; University of Illinois at Urbana-Champaign, Roger Adams Laboratory, 600 S. Mathews Ave., Urbana IL, 61801, USA.
Organizational Affiliation: