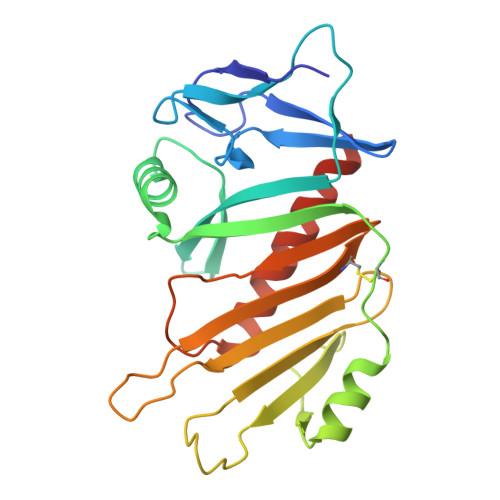
Structural basis of the interaction between the putative adhesion-involved and iron-regulated FrpD and FrpC proteins of Neisseria meningitidis.
Sviridova, E., Rezacova, P., Bondar, A., Veverka, V., Novak, P., Schenk, G., Svergun, D.I., Kuta Smatanova, I., Bumba, L.(2017) Sci Rep 7: 40408-40408
- PubMed: 28084396 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/srep40408
- Primary Citation Related Structures:
5EDF, 5EDJ - PubMed Abstract:
The iron-regulated protein FrpD from Neisseria meningitidis is an outer membrane lipoprotein that interacts with very high affinity (K d ~ 0.2 nM) with the N-terminal domain of FrpC, a Type I-secreted protein from the Repeat in ToXin (RTX) protein family. In the presence of Ca 2+ , FrpC undergoes Ca 2+ -dependent protein trans-splicing that includes an autocatalytic cleavage of the Asp 414 -Pro 415 peptide bond and formation of an Asp 414 -Lys isopeptide bond. Here, we report the high-resolution structure of FrpD and describe the structure-function relationships underlying the interaction between FrpD and FrpC 1-414 . We identified FrpD residues involved in FrpC 1-414 binding, which enabled localization of FrpD within the low-resolution SAXS model of the FrpD-FrpC 1-414 complex. Moreover, the trans-splicing activity of FrpC resulted in covalent linkage of the FrpC 1-414 fragment to plasma membrane proteins of epithelial cells in vitro, suggesting that formation of the FrpD-FrpC 1-414 complex may be involved in the interaction of meningococci with the host cell surface.
- Faculty of Science, University of South Bohemia Ceske Budejovice, Branisovska 1760, 37005 Ceske Budejovice, Czech Republic.
Organizational Affiliation: