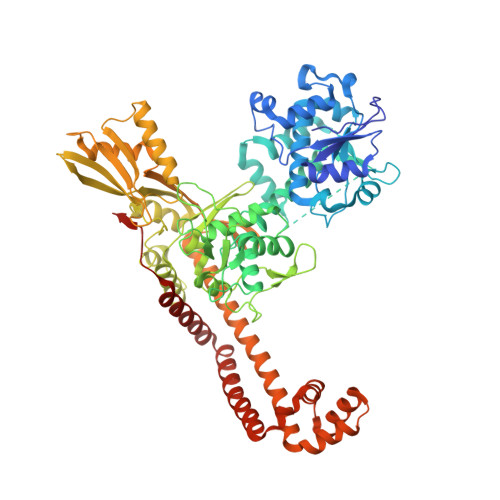
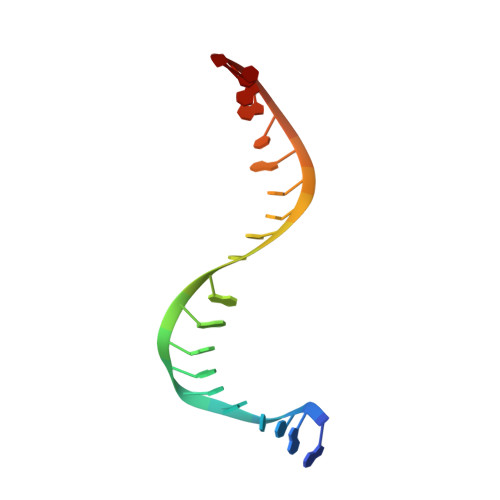
Tricyclic 1,5-naphthyridinone oxabicyclooctane-linked novel bacterial topoisomerase inhibitors as broad-spectrum antibacterial agents-SAR of left-hand-side moiety (Part-2).
Singh, S.B., Kaelin, D.E., Wu, J., Miesel, L., Tan, C.M., Black, T., Nargund, R., Meinke, P.T., Olsen, D.B., Lagrutta, A., Lu, J., Patel, S., Rickert, K.W., Smith, R.F., Soisson, S., Sherer, E., Joyce, L.A., Wei, C., Peng, X., Wang, X., Fukuda, H., Kishii, R., Takei, M., Takano, H., Shibasaki, M., Yajima, M., Nishimura, A., Shibata, T., Fukuda, Y.(2015) Bioorg Med Chem Lett 25: 1831-1835
- PubMed: 25851938 Search on PubMed
- DOI: https://doi.org/10.1016/j.bmcl.2015.03.044
- Primary Citation Related Structures:
5BS3 - PubMed Abstract:
Novel bacterial topoisomerase inhibitors (NBTIs) represent a new class of broad-spectrum antibacterial agents targeting bacterial Gyrase A and ParC and have potential utility in combating antibiotic resistance. A series of novel oxabicyclooctane-linked NBTIs with new tricyclic-1,5-naphthyridinone left hand side moieties have been described. Compounds with a (R)-hydroxy-1,5-naphthyridinone moiety (7) showed potent antibacterial activity (e.g., Staphylococcus aureus MIC 0.25 μg/mL), acceptable Gram-positive and Gram-negative spectrum with rapidly bactericidal activity. The compound 7 showed intravenous and oral efficacy (ED50) at 3.2 and 27 mg/kg doses, respectively, in a murine model of bacteremia. Most importantly they showed significant attenuation of functional hERG activity (IC50 >170 μM). In general, lower logD attenuated hERG activity but also reduced Gram-negative activity. The co-crystal structure of a hydroxy-tricyclic NBTI bound to a DNA-gyrase complex exhibited a binding mode that show enantiomeric preference for R isomer and explains the activity and SAR. The discovery, synthesis, SAR and X-ray crystal structure of the left-hand-side tricyclic 1,5-naphthyridinone based oxabicyclooctane linked NBTIs are described.
- Merck Research Laboratories, Kenilworth, NJ 07033, United States. Electronic address: sheo.singh.215@gmail.com.
Organizational Affiliation: