Mutation of the conserved serine in two PurE classes
Sullivan, K.L., Kappock, T.J.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| N5-carboxyaminoimidazole ribonucleotide mutase | 183 | Acetobacter aceti 1023 | Mutation(s): 1 Gene Names: purE, AZ09_02690 EC: 5.4.99.18 | 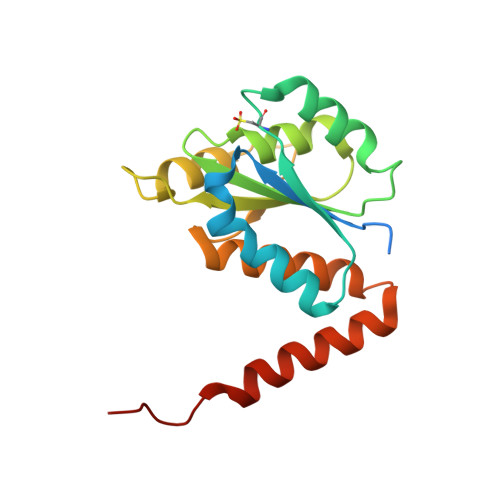 | |
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| N5-carboxyaminoimidazole ribonucleotide mutase | 183 | Acetobacter aceti 1023 | Mutation(s): 1 Gene Names: purE, AZ09_02690 EC: 5.4.99.18 | 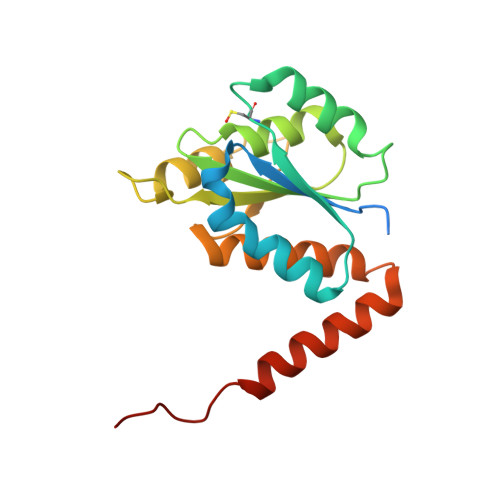 | |
| Ligands 4 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| SO4 Download:Ideal Coordinates CCD File | H [auth B] | SULFATE ION O4 S QAOWNCQODCNURD-UHFFFAOYSA-L |  | ||
| EDO Download:Ideal Coordinates CCD File | E [auth B], G [auth B] | 1,2-ETHANEDIOL C2 H6 O2 LYCAIKOWRPUZTN-UHFFFAOYSA-N |  | ||
| ACT Download:Ideal Coordinates CCD File | C [auth A], F [auth B] | ACETATE ION C2 H3 O2 QTBSBXVTEAMEQO-UHFFFAOYSA-M |  | ||
| CL Download:Ideal Coordinates CCD File | D [auth A] | CHLORIDE ION Cl VEXZGXHMUGYJMC-UHFFFAOYSA-M |  | ||
| Modified Residues 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| OCS Query on OCS | A | L-PEPTIDE LINKING | C3 H7 N O5 S |  | CYS |
| CSX Query on CSX | B | L-PEPTIDE LINKING | C3 H7 N O3 S |  | CYS |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 99.584 | α = 90 |
| b = 99.584 | β = 90 |
| c = 164.702 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |
| PHASER | phasing |