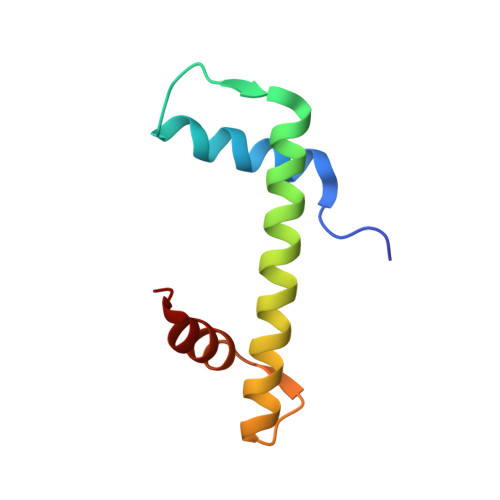
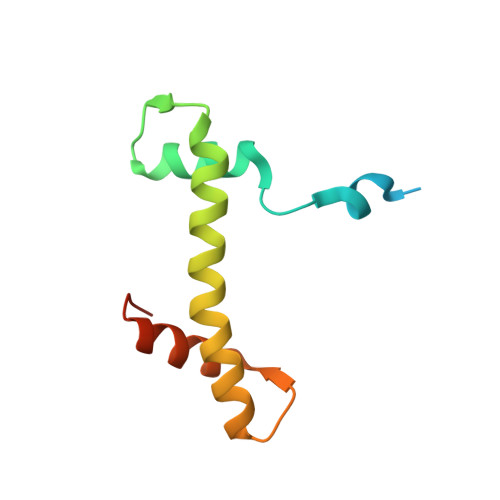
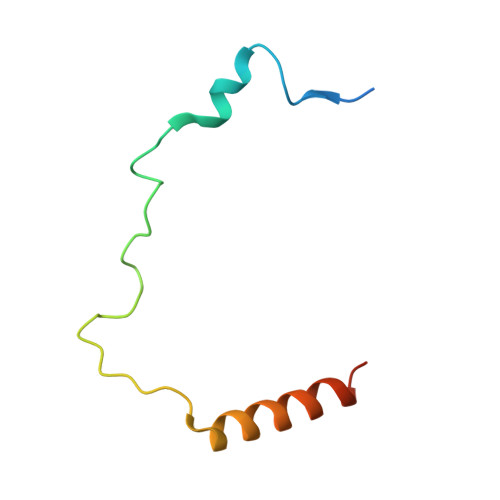
A unique binding mode enables MCM2 to chaperone histones H3-H4 at replication forks.
Huang, H., Strmme, C.B., Saredi, G., Hodl, M., Strandsby, A., Gonzalez-Aguilera, C., Chen, S., Groth, A., Patel, D.J.(2015) Nat Struct Mol Biol 22: 618-626
- PubMed: 26167883 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/nsmb.3055
- Primary Citation Related Structures:
5BNV, 5BNX, 5BO0 - PubMed Abstract:
During DNA replication, chromatin is reassembled by recycling of modified old histones and deposition of new ones. How histone dynamics integrates with DNA replication to maintain genome and epigenome information remains unclear. Here, we reveal how human MCM2, part of the replicative helicase, chaperones histones H3-H4. Our first structure shows an H3-H4 tetramer bound by two MCM2 histone-binding domains (HBDs), which hijack interaction sites used by nucleosomal DNA. Our second structure reveals MCM2 and ASF1 cochaperoning an H3-H4 dimer. Mutational analyses show that the MCM2 HBD is required for MCM2-7 histone-chaperone function and normal cell proliferation. Further, we show that MCM2 can chaperone both new and old canonical histones H3-H4 as well as H3.3 and CENPA variants. The unique histone-binding mode of MCM2 thus endows the replicative helicase with ideal properties for recycling histones genome wide during DNA replication.
- Structural Biology Program, Memorial Sloan-Kettering Cancer Center, New York, New York, USA.
Organizational Affiliation: