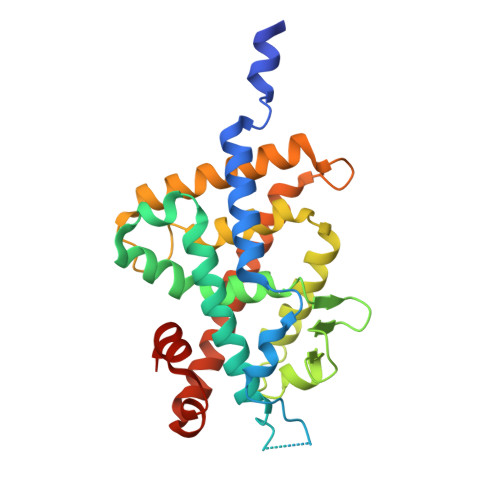
Helix12-Stabilization Antagonist of Vitamin D Receptor
Kato, A., Itoh, T., Anami, Y., Egawa, D., Yamamoto, K.(2016) Bioconjug Chem 27: 1750-1761
- PubMed: 27294600 Search on PubMed
- DOI: https://doi.org/10.1021/acs.bioconjchem.6b00246
- Primary Citation Related Structures:
5B5B - PubMed Abstract:
To develop strong vitamin D receptor (VDR) antagonists and reveal their antagonistic mechanism, we designed and synthesized vitamin D analogues with bulky side chains based on the "active antagonist" concept in which antagonist prevents helix 12 (H12) folding. Of the synthesized analogues, compounds 3a and 3b showed strong antagonistic activity. Dynamic hydrogen/deuterium exchange coupled with mass spectrometry (HDX-MS) and static X-ray crystal structure analyses indicated that compound 3a stabilizes H11-H12 but displaces H6-H7 so that 3a is a novel rather than "active" or "passive" type of antagonist. We classified 3a as a third type of antagonist and called it "H11-H12 stabilization antagonist". HDX-MS analysis indicated that antagonist 3b is an "active" antagonist. To date there are no reports relating to nuclear receptor antagonist that strongly stabilizes H12. In this study, we found first VDR antagonist that stabilizes H12 and we showed that antagonistic mechanism is diverse depending on each antagonist structure. Additionally, HDX-MS was proven to be very useful for investigations of protein structure alterations resulting from ligand binding.
- Laboratory of Drug Design and Medicinal Chemistry, Showa Pharmaceutical University , 3-3165 Higashi-Tamagawagakuen, Machida, Tokyo 194-8543, Japan.
Organizational Affiliation: