Apo- and Antagonist-Binding Structures of Vitamin D Receptor Ligand-Binding Domain Revealed by Hybrid Approach Combining Small-Angle X-ray Scattering and Molecular Dynamics
Anami, Y., Shimizu, N., Ekimoto, T., Egawa, D., Itoh, T., Ikeguchi, M., Yamamoto, K.(2016) J Med Chem 59: 7888-7900
- PubMed: 27535484 Search on PubMed
- DOI: https://doi.org/10.1021/acs.jmedchem.6b00682
- Primary Citation Related Structures:
5B41 - PubMed Abstract:
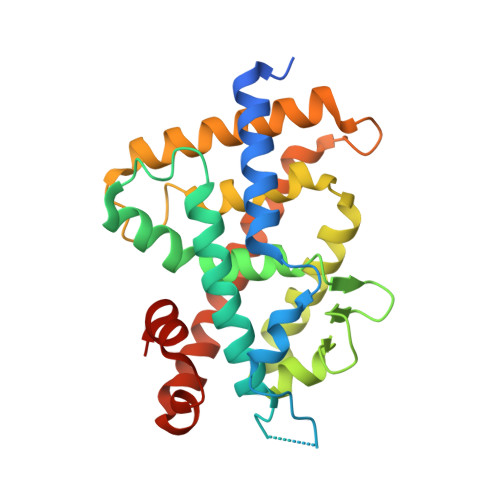
Vitamin D receptor (VDR) controls the expression of numerous genes through the conformational change caused by binding 1α,25-dihydroxyvitamin D3. Helix 12 in the ligand-binding domain (LBD) is key to regulating VDR activation. The structures of apo VDR-LBD and the VDR-LBD/antagonist complex are unclear. Here, we reveal their unprecedented structures in solution using a hybrid method combining small-angle X-ray scattering and molecular dynamics simulations. In apo rat VDR-LBD, helix 12 is partially unraveled, and it is positioned around the canonical active position and fluctuates. Helix 11 greatly bends toward the outside at Q396, creating a kink. In the rat VDR-LBD/antagonist complex, helix 12 does not generate the activation function 2 surface, and loop 11-12 is remarkably flexible compared to that in the apo rat VDR-LBD. On the basis of these structural insights, we propose a "folding-door model" to describe the mechanism of agonism/antagonism of VDR-LBD.
- Laboratory of Drug Design and Medicinal Chemistry, Showa Pharmaceutical University , 3-3165 Higashi-Tamagawagakuen, Machida, Tokyo 194-8543, Japan.
Organizational Affiliation: