New Insights into the Design of Inhibitors Targeted for Parasitic Anaerobic Energy Metabolism
Harada, S., Shiba, T., Sato, D., Yamamoto, A., Nagahama, M., Yone, A., Inaoka, D.K., Sakamoto, K., Inoue, M., Honma, T., Kita, K.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Succinate dehydrogenase flavoprotein | 645 | Ascaris suum | Mutation(s): 0 EC: 1.3.5.1 Membrane Entity: Yes | 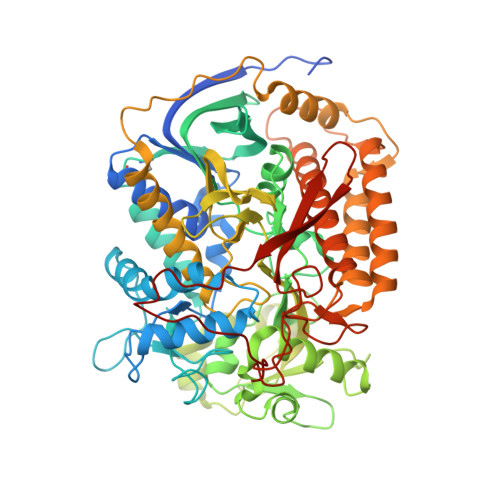 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q33862 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Succinate dehydrogenase [ubiquinone] iron-sulfur subunit, mitochondrial | 282 | Ascaris suum | Mutation(s): 0 EC: 1.3.5.1 Membrane Entity: Yes | 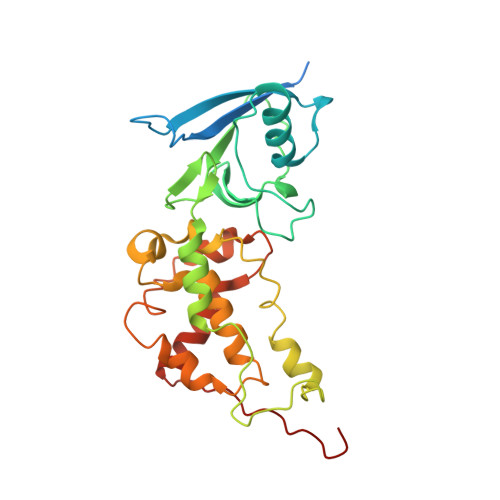 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | O44074 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Cytochrome b-large subunit | 188 | Ascaris suum | Mutation(s): 0 Membrane Entity: Yes | 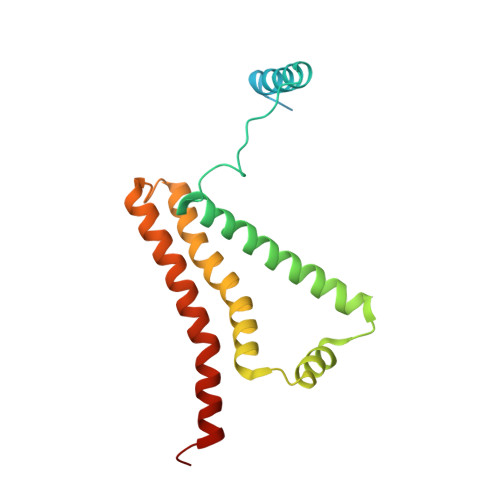 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P92506 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 4 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Succinate dehydrogenase [ubiquinone] cytochrome b small subunit, mitochondrial | 156 | Ascaris suum | Mutation(s): 0 Membrane Entity: Yes | 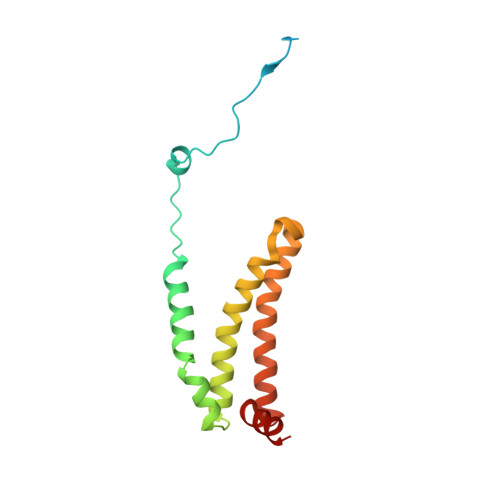 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P92507 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 7 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| FAD Download:Ideal Coordinates CCD File | J [auth A], Q [auth E] | FLAVIN-ADENINE DINUCLEOTIDE C27 H33 N9 O15 P2 VWWQXMAJTJZDQX-UYBVJOGSSA-N |  | ||
| HEM Download:Ideal Coordinates CCD File | N [auth C], V [auth G] | PROTOPORPHYRIN IX CONTAINING FE C34 H32 Fe N4 O4 KABFMIBPWCXCRK-RGGAHWMASA-L |  | ||
| FD8 Download:Ideal Coordinates CCD File | O [auth C], U [auth F] | N-[3-(pentafluorophenoxy)phenyl]-2-(trifluoromethyl)benzamide C20 H9 F8 N O2 ZTJJBXFGRJEBCQ-UHFFFAOYSA-N |  | ||
| SF4 Download:Ideal Coordinates CCD File | L [auth B], S [auth F] | IRON/SULFUR CLUSTER Fe4 S4 LJBDFODJNLIPKO-UHFFFAOYSA-N |  | ||
| F3S Download:Ideal Coordinates CCD File | M [auth B], T [auth F] | FE3-S4 CLUSTER Fe3 S4 FCXHZBQOKRZXKS-UHFFFAOYSA-N |  | ||
| FES Download:Ideal Coordinates CCD File | K [auth B], R [auth F] | FE2/S2 (INORGANIC) CLUSTER Fe2 S2 NIXDOXVAJZFRNF-UHFFFAOYSA-N |  | ||
| MLI Download:Ideal Coordinates CCD File | I [auth A], P [auth E] | MALONATE ION C3 H2 O4 OFOBLEOULBTSOW-UHFFFAOYSA-L |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 123.614 | α = 90 |
| b = 125.483 | β = 90 |
| c = 219.67 | γ = 90 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| HKL-2000 | data processing |
| HKL-2000 | data scaling |
| MOLREP | phasing |