Crystal structure of a putative ssRNA endonuclease Cas2, CRISPR adaptation protein from E.coli
Nocek, B., Skarina, T., Brown, G., Yakunin, A., Joachimiak, A.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| CRISPR-associated endoribonuclease Cas2 | 94 | Escherichia coli str. K-12 substr. MG1655 | Mutation(s): 0 Gene Names: ygbF, cas2, b2754, JW5438 EC: 3.1 | 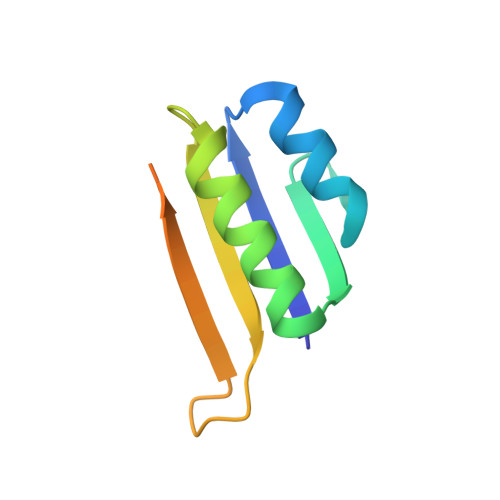 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P45956 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| PEG Download:Ideal Coordinates CCD File | C [auth B] | DI(HYDROXYETHYL)ETHER C4 H10 O3 MTHSVFCYNBDYFN-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 30.048 | α = 90 |
| b = 66.009 | β = 90 |
| c = 69.972 | γ = 90 |
| Software Name | Purpose |
|---|---|
| SBC-Collect | data collection |
| CCP4 | model building |
| MOLREP | phasing |
| REFMAC | refinement |
| HKL-3000 | data reduction |
| HKL-3000 | data scaling |
| CCP4 | phasing |