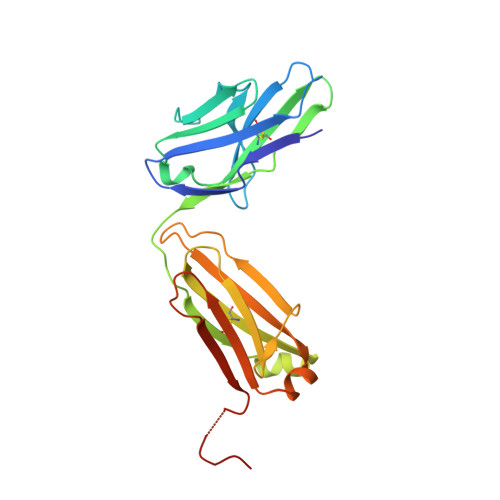
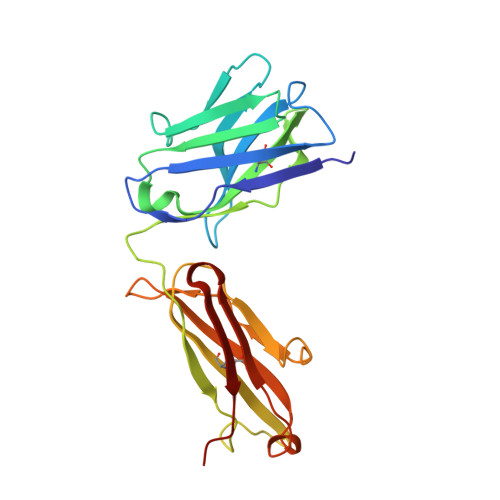
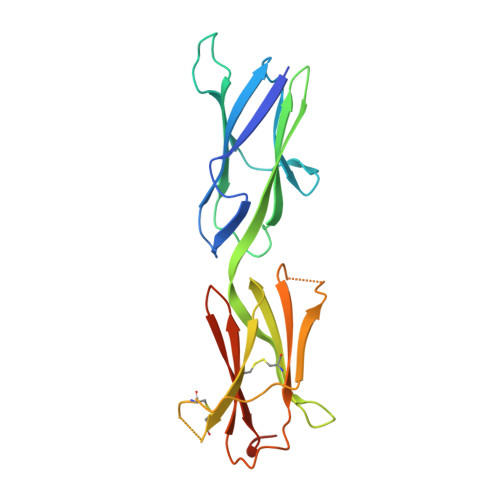
Structural basis for KIT receptor tyrosine kinase inhibition by antibodies targeting the D4 membrane-proximal region.
Reshetnyak, A.V., Nelson, B., Shi, X., Boggon, T.J., Pavlenco, A., Mandel-Bausch, E.M., Tome, F., Suzuki, Y., Sidhu, S.S., Lax, I., Schlessinger, J.(2013) Proc Natl Acad Sci U S A 110: 17832-17837
- PubMed: 24127596 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1073/pnas.1317118110
- Primary Citation Related Structures:
4K94, 4K9E - PubMed Abstract:
Somatic oncogenic mutations in the receptor tyrosine kinase KIT function as major drivers of gastrointestinal stromal tumors and a subset of acute myeloid leukemia, melanoma, and other cancers. Although treatment of these cancers with tyrosine kinase inhibitors shows dramatic responses and durable disease control, drug resistance followed by clinical progression of disease eventually occurs in virtually all patients. In this report, we describe inhibitory KIT antibodies that bind to the membrane-proximal Ig-like D4 of KIT with significant overlap with an epitope in D4 that mediates homotypic interactions essential for KIT activation. Crystal structures of the anti-KIT antibody in complex with KIT D4 and D5 allowed design of affinity-matured libraries that were used to isolate variants with increased affinity and efficacy. Isolated antibodies showed KIT inhibition together with suppression of cell proliferation driven by ligand-stimulated WT or constitutively activated oncogenic KIT mutant. These antibodies represent a unique therapeutic approach and a step toward the development of "naked" or toxin-conjugated KIT antibodies for the treatment of KIT-driven cancers.
- Department of Pharmacology, Yale University School of Medicine, New Haven, CT 06520.
Organizational Affiliation: