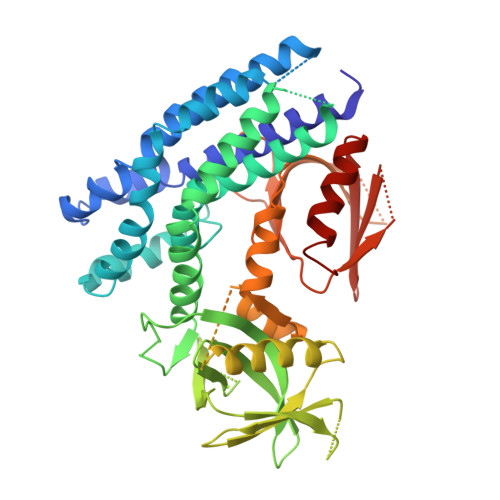
Structural Basis for Autoinhibition of the Guanine Nucleotide Exchange Factor FARP2.
He, X., Kuo, Y.C., Rosche, T.J., Zhang, X.(2013) Structure 21: 355-364
- PubMed: 23375260 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.str.2013.01.001
- Primary Citation Related Structures:
4GYV, 4GZU, 4H6Y - PubMed Abstract:
FARP2 is a Dbl-family guanine nucleotide exchange factor (GEF) that contains a 4.1, ezrin, radixin and moesin (FERM) domain, a Dbl-homology (DH) domain and two pleckstrin homology (PH) domains. FARP2 activates Rac1 or Cdc42 in response to upstream signals, thereby regulating processes such as neuronal axon guidance and bone homeostasis. How the GEF activity of FARP2 is regulated remained poorly understood. We have determined the crystal structures of the catalytic DH domain and the DH-PH-PH domains of FARP2. The structures reveal an auto-inhibited conformation in which the GEF substrate-binding site is blocked collectively by the last helix in the DH domain and the two PH domains. This conformation is stabilized by multiple interactions among the domains and two well-structured inter-domain linkers. Our cell-based activity assays confirm the suppression of the FARP2 GEF activity by these auto-inhibitory elements.
- Department of Pharmacology, University of Texas Southwestern Medical Center, Dallas, TX 75063, USA.
Organizational Affiliation: