A mutant O-GlcNAcase as a probe to reveal global dynamics of protein O-GlcNAcylation during Drosophila embryonic development.
Mariappa, D., Selvan, N., Borodkin, V.S., Alonso, J., Ferenbach, A.T., Shepherd, C., Navratilova, I.H., van Aalten, D.M.(2015) Biochem J 470: 255-262
- PubMed: 26348912 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1042/BJ20150610
- Primary Citation Related Structures:
4ZXL - PubMed Abstract:
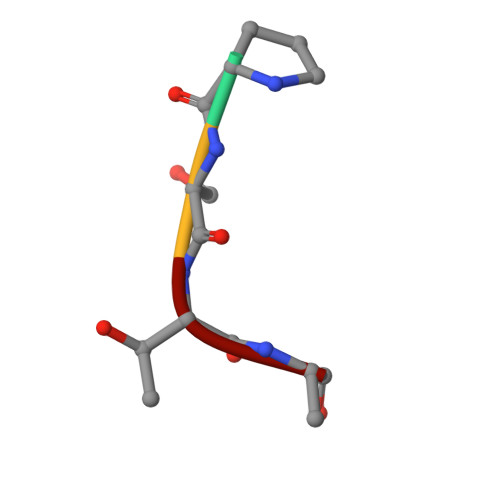
O-GlcNAcylation is a reversible type of serine/threonine glycosylation on nucleocytoplasmic proteins in metazoa. Various genetic approaches in several animal models have revealed that O-GlcNAcylation is essential for embryogenesis. However, the dynamic changes in global O-GlcNAcylation and the underlying mechanistic biology linking them to embryonic development is not understood. One of the limiting factors towards characterizing changes in O-GlcNAcylation has been the limited specificity of currently available tools to detect this modification. In the present study, harnessing the unusual properties of an O-GlcNAcase (OGA) mutant that binds O-GlcNAc (O-N-acetylglucosamine) sites with nanomolar affinity, we uncover changes in protein O-GlcNAcylation as a function of Drosophila development.
- MRC Protein Phosphorylation and Ubiquitylation Unit, College of Life Sciences, University of Dundee, Dow Street, Dundee, DD1 5EH, United Kingdom.
Organizational Affiliation: