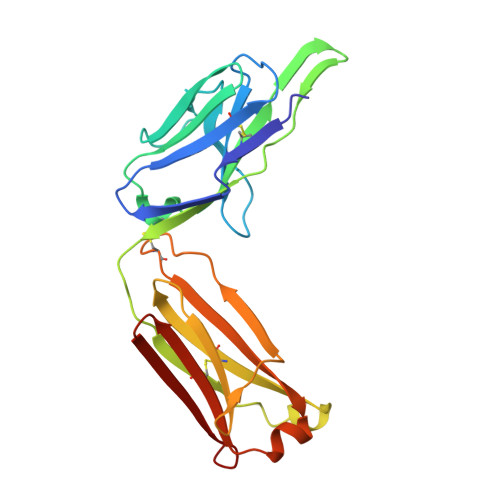
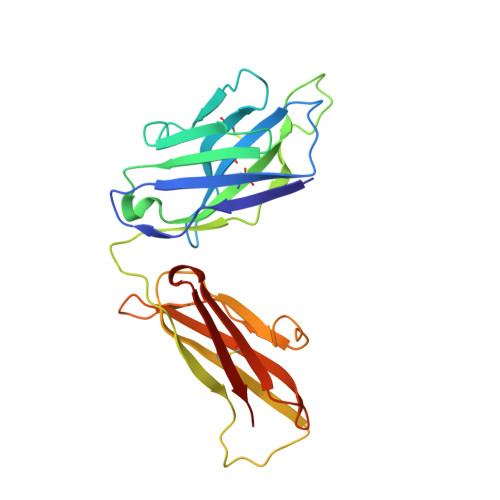
Structural analysis of a novel rabbit monoclonal antibody R53 targeting an epitope in HIV-1 gp120 C4 region critical for receptor and co-receptor binding.
Pan, R., Chen, Y., Vaine, M., Hu, G., Wang, S., Lu, S., Kong, X.P.(2015) Emerg Microbes Infect 4: e44-e44
- PubMed: 26251831 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/emi.2015.44
- Primary Citation Related Structures:
4ZTO, 4ZTP - PubMed Abstract:
The fourth conserved region (C4) in the HIV-1 envelope glycoprotein (Env) gp120 is a structural element that is important for its function, as it binds to both the receptor CD4 and the co-receptor CCR5/CXCR4. It has long been known that this region is highly immunogenic and that it harbors B-cell as well as T-cell epitopes. It is the target of a number of antibodies in animal studies, which are called CD4-blockers. However, the mechanism by which the virus shields itself from such antibody responses is not known. Here, we determined the crystal structure of R53 in complex with its epitope peptide using a novel anti-C4 rabbit monoclonal antibody R53. Our data show that although the epitope of R53 covers a highly conserved sequence (433)AMYAPPI(439), it is not available in the gp120 trimer and in the CD4-bound conformation. Our results suggest a masking mechanism to explain how HIV-1 protects this critical region from the human immune system.
- Departments of Biochemistry and Molecular Pharmacology, New York University School of Medicine , New York, NY 10016, USA.
Organizational Affiliation: