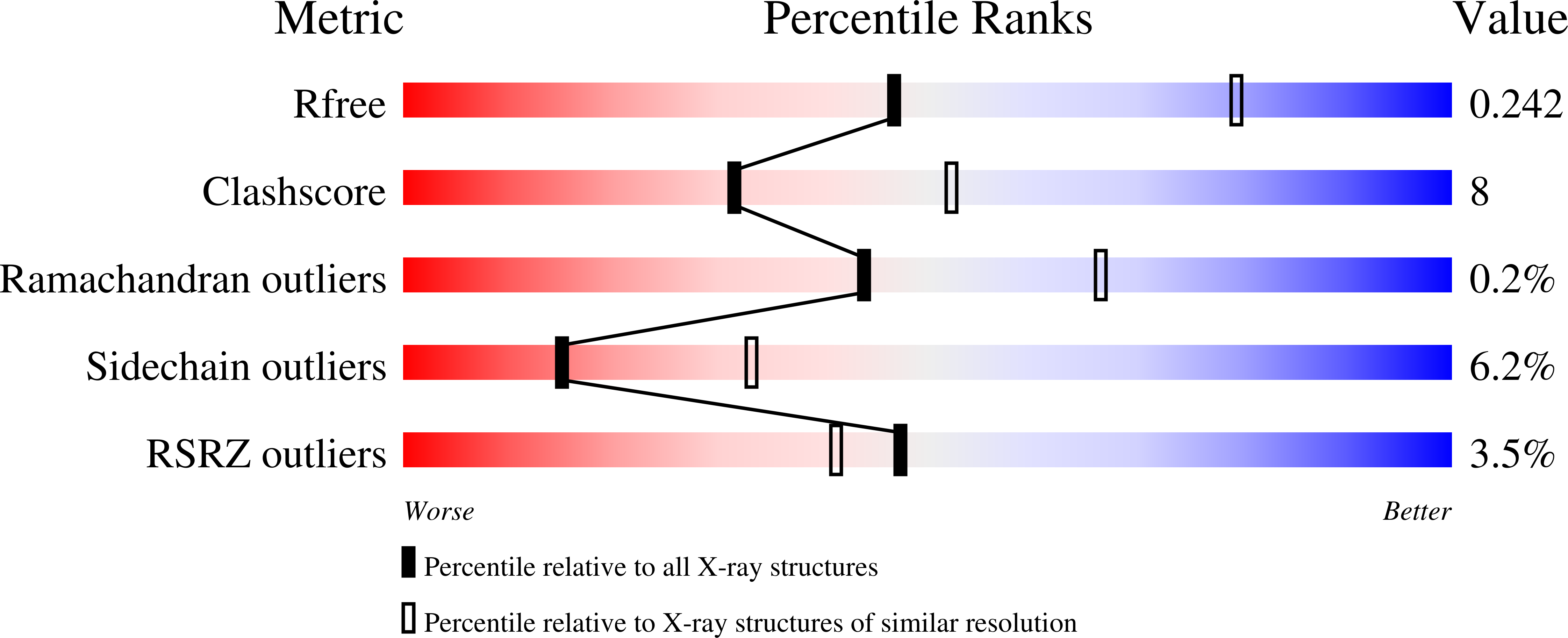
Evaluation of candidate vaccine approaches for MERS-CoV
Joyce, M.G.(2015) Nat Commun 6: 7712
- PubMed: 26218507 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/ncomms8712
- Primary Citation Related Structures:
4ZPT, 4ZPV, 4ZPW - PubMed Abstract:
The emergence of Middle East respiratory syndrome coronavirus (MERS-CoV) as a cause of severe respiratory disease highlights the need for effective approaches to CoV vaccine development. Efforts focused solely on the receptor-binding domain (RBD) of the viral Spike (S) glycoprotein may not optimize neutralizing antibody (NAb) responses. Here we show that immunogens based on full-length S DNA and S1 subunit protein elicit robust serum-neutralizing activity against several MERS-CoV strains in mice and non-human primates. Serological analysis and isolation of murine monoclonal antibodies revealed that immunization elicits NAbs to RBD and, non-RBD portions of S1 and S2 subunit. Multiple neutralization mechanisms were demonstrated by solving the atomic structure of a NAb-RBD complex, through sequencing of neutralization escape viruses and by constructing MERS-CoV S variants for serological assays. Immunization of rhesus macaques confers protection against MERS-CoV-induced radiographic pneumonia, as assessed using computerized tomography, supporting this strategy as a promising approach for MERS-CoV vaccine development.
- Vaccine Research Center, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Bethesda, Maryland 20892, USA.
Organizational Affiliation: