Structural basis for calcium regulation of myosin 5 motor function
Shen, M., Zheng, S., Zhang, W., Lu, Z., Ye, K., Li, X.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Unconventional myosin-Va | 839 | Mus musculus | Mutation(s): 0 Gene Names: Myo5a, Dilute | 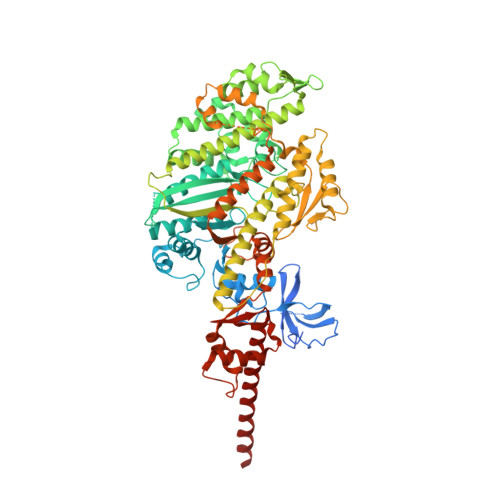 | |
UniProt & NIH Common Fund Data Resources | |||||
IMPC: MGI:105976 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q99104 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Calmodulin | 149 | Mus musculus | Mutation(s): 0 Gene Names: Calm1, Calm, Cam, Cam1, Calm2, Cam2, Camb, Calm3, Cam3, Camc | 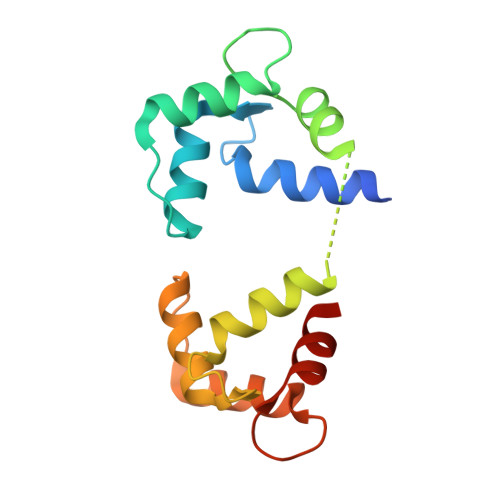 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P0DP26 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| CA Download:Ideal Coordinates CCD File | C [auth B], D [auth B], E [auth B], F [auth B] | CALCIUM ION Ca BHPQYMZQTOCNFJ-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 56.079 | α = 90 |
| b = 99.455 | β = 90 |
| c = 248.159 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |
| PHASER | phasing |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Basic Research Program of China | China | 2013CB932802 |
| National Basic Research Program of China | China | 2012CB114102 |
| National Natural Science Foundation of China | China | 31171367 |
| National Natural Science Foundation of China | China | 31470791 |
| National Natural Science Foundation of China | China | 31325007 |