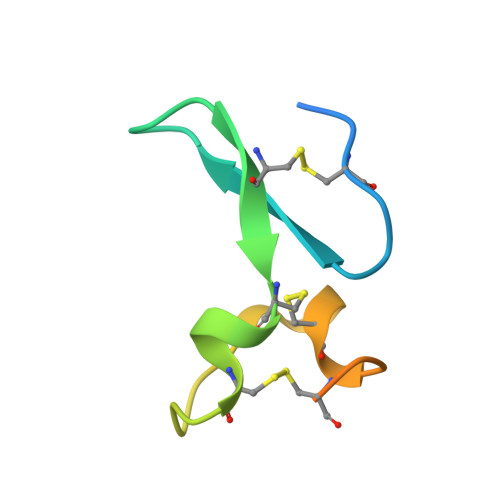
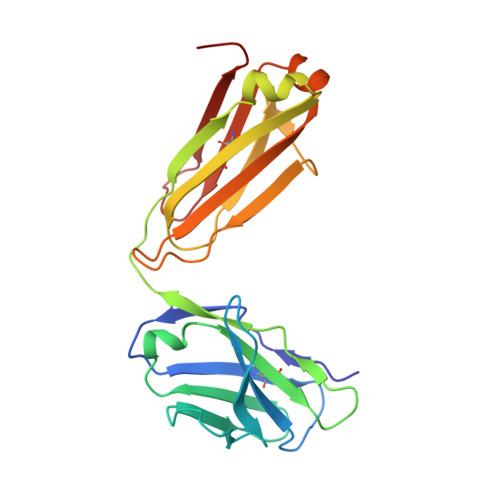
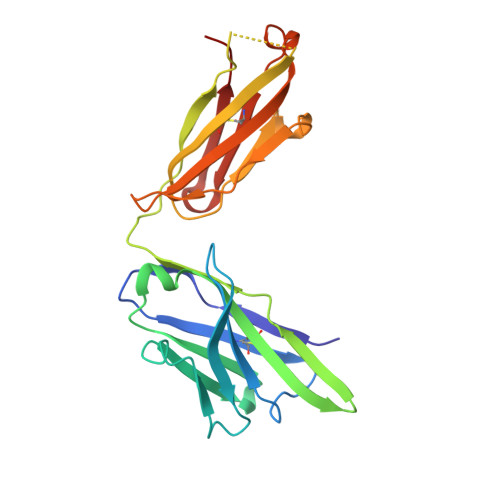
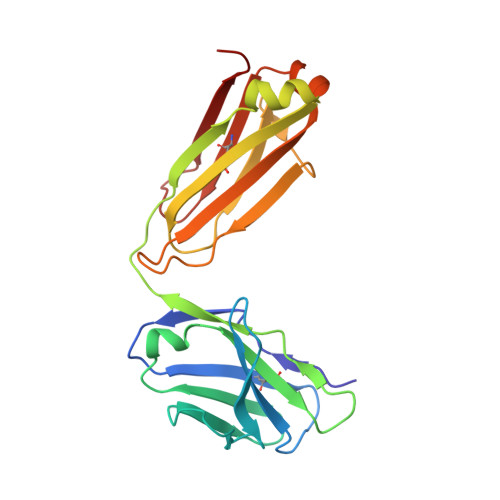
Potent anti-tumor response by targeting B cell maturation antigen (BCMA) in a mouse model of multiple myeloma.
Oden, F., Marino, S.F., Brand, J., Scheu, S., Kriegel, C., Olal, D., Takvorian, A., Westermann, J., Yilmaz, B., Hinz, M., Daumke, O., Hopken, U.E., Muller, G., Lipp, M.(2015) Mol Oncol 9: 1348-1358
- PubMed: 25953704 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1016/j.molonc.2015.03.010
- Primary Citation Related Structures:
4ZFO - PubMed Abstract:
Multiple myeloma (MM) is an aggressive incurable plasma cell malignancy with a median life expectancy of less than seven years. Antibody-based therapies have demonstrated substantial clinical benefit for patients with hematological malignancies, particular in B cell Non-Hodgkin's lymphoma. The lack of immunotherapies specifically targeting MM cells led us to develop a human-mouse chimeric antibody directed against the B cell maturation antigen (BCMA), which is almost exclusively expressed on plasma cells and multiple myeloma cells. The high affinity antibody blocks the binding of the native ligands APRIL and BAFF to BCMA. This finding is rationalized by the high resolution crystal structure of the Fab fragment in complex with the extracellular domain of BCMA. Most importantly, the antibody effectively depletes MM cells in vitro and in vivo and substantially prolongs tumor-free survival under therapeutic conditions in a xenograft mouse model. A BCMA-antibody-based therapy is therefore a promising option for the effective treatment of multiple myeloma and autoimmune diseases.
- Max-Delbrück-Center of Molecular Medicine (MDC), Department of Tumor Genetics and Immunogenetics, Robert-Rössle-Strasse 10, 13125 Berlin, Germany. Electronic address: felix.oden@mdc-berlin.de.
Organizational Affiliation: