Crystal Structure of a GNAT family Acetyltransferase from Brucella melitensis in complex with Acetyl-CoA
SSGCID, Dranow, D.M., Trakaki, T., Lorimer, D., Edwards, T.E.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Acetyltransferase | 172 | Brucella abortus str. 2308 A | Mutation(s): 0 Gene Names: BAAA_2000484 | 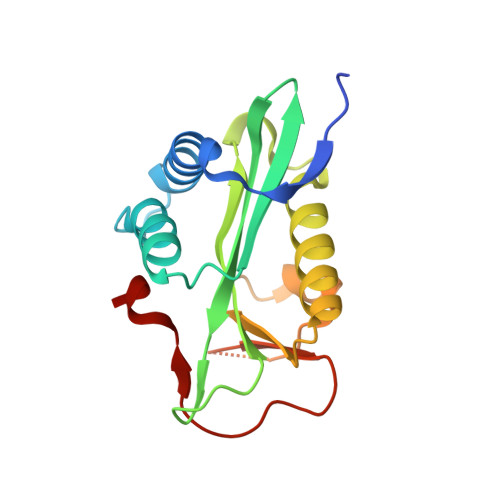 | |
| Ligands 2 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| ACO Download:Ideal Coordinates CCD File | B [auth A] | ACETYL COENZYME *A C23 H38 N7 O17 P3 S ZSLZBFCDCINBPY-ZSJPKINUSA-N |  | ||
| GOL Download:Ideal Coordinates CCD File | C [auth A] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 29.81 | α = 90 |
| b = 57.11 | β = 96.53 |
| c = 44.06 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| PDB_EXTRACT | data extraction |
| XDS | data reduction |
| XSCALE | data scaling |
| BALBES | phasing |