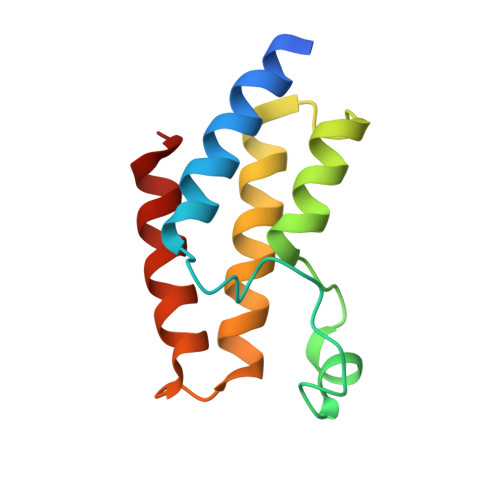
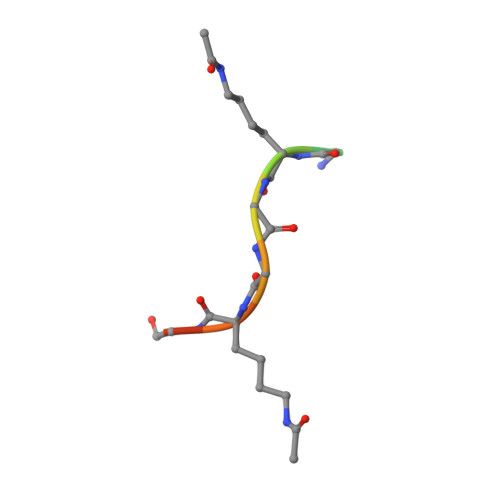
A Subset of Human Bromodomains Recognizes Butyryllysine and Crotonyllysine Histone Peptide Modifications.
Flynn, E.M., Huang, O.W., Poy, F., Oppikofer, M., Bellon, S.F., Tang, Y., Cochran, A.G.(2015) Structure 23: 1801-1814
- PubMed: 26365797 Search on PubMed
- DOI: https://doi.org/10.1016/j.str.2015.08.004
- Primary Citation Related Structures:
4YY4, 4YY6, 4YYD, 4YYG, 4YYH, 4YYI, 4YYJ, 4YYK, 4YYM, 4YYN - PubMed Abstract:
Bromodomains are epigenetic readers that are recruited to acetyllysine residues in histone tails. Recent studies have identified non-acetyl acyllysine modifications, raising the possibility that these might be read by bromodomains. Profiling the nearly complete human bromodomain family revealed that while most human bromodomains bind only the shorter acetyl and propionyl marks, the bromodomains of BRD9, CECR2, and the second bromodomain of TAF1 also recognize the longer butyryl mark. In addition, the TAF1 second bromodomain is capable of binding crotonyl marks. None of the human bromodomains tested binds succinyl marks. We characterized structurally and biochemically the binding to different acyl groups, identifying bromodomain residues and structural attributes that contribute to specificity. These studies demonstrate a surprising degree of plasticity in some human bromodomains but no single factor controlling specificity across the family. The identification of candidate butyryl- and crotonyllysine readers supports the idea that these marks could have specific physiological functions.
- Department of Early Discovery Biochemistry, Genentech, Inc., 1 DNA Way, South San Francisco, CA 94080, USA.
Organizational Affiliation: