Structure of hemagglutinin from a H6N1 influenza virus (A/chicken/Taiwan/A2837/2013)
Wang, F., Qi, J., Bi, Y., Zhang, W., Wang, M., Wang, M., Liu, J., Yan, J., Shi, Y., Gao, G.F.To be published.
Experimental Data Snapshot
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| HA1 | 325 | H6N1 subtype | Mutation(s): 0 | 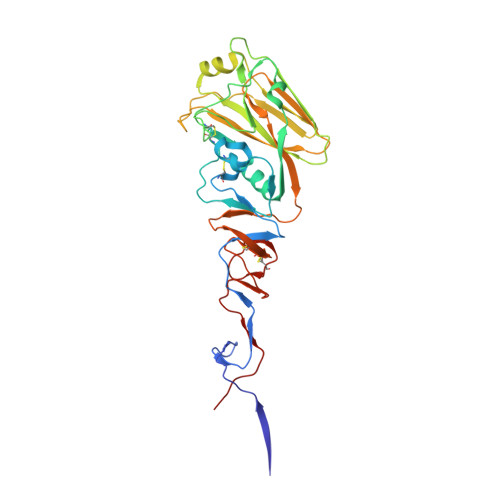 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A0A0J9X268 | ||||
Glycosylation | |||||
| Glycosylation Sites: 2 | |||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| HA2 | 164 | H6N1 subtype | Mutation(s): 0 | 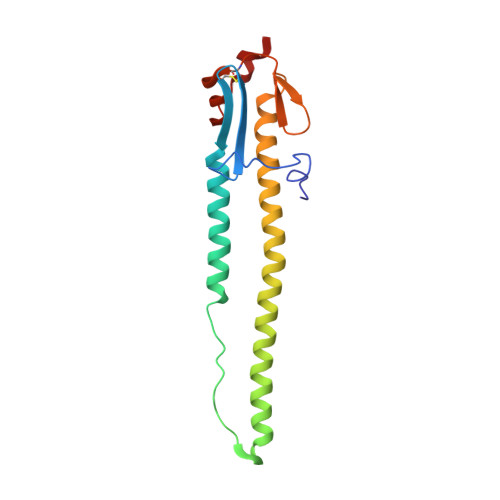 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A0A0J9X267 | ||||
Glycosylation | |||||
| Glycosylation Sites: 1 | |||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Length | 2D Diagram | Glycosylation | D Interactions |
| 2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose | E, G | 2 |  | N-Glycosylation | |
Glycosylation Resources | |||||
GlyTouCan: G42666HT GlyCosmos: G42666HT GlyGen: G42666HT | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| NAG Download:Ideal Coordinates CCD File | I [auth A], J [auth B], K [auth C], L [auth D] | 2-acetamido-2-deoxy-beta-D-glucopyranose C8 H15 N O6 OVRNDRQMDRJTHS-FMDGEEDCSA-N |  | ||
Entity ID: 4 | |||||
|---|---|---|---|---|---|
| ID | Chains | Name | Type/Class | 2D Diagram | 3D Interactions |
| PRD_900067 Query on PRD_900067 | F, H | 3'-sialyl-N-acetyllactosamine | Oligosaccharide / Substrate analog |  |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 96.772 | α = 90 |
| b = 96.772 | β = 90 |
| c = 132.506 | γ = 120 |
| Software Name | Purpose |
|---|---|
| HKL-2000 | data processing |
| PHASER | phasing |
| Coot | model building |
| PHENIX | refinement |
| SCALA | data reduction |
| SCALEPACK | data scaling |
| Funding Organization | Location | Grant Number |
|---|---|---|
| China Ministry of Science and Technology National 973 Project | China | 2011CB504703 |
| Intramural Special Grant for Influenza Virus Research from the Chinese Academy of Sciences | China | KJZD-EW-L09 |
| Intramural Special Grant for Strategic Priority Research Program of the Chinese Academy of Sciences | China | XDB08020100 |
| National Natural Science Foundation of China | China | 31402196 |
| China National Grand S&T Special Project | China | 2014ZX10004002 |