Structural basis of human galectin-1 inhibition with Ki values in the micro- to nanomolar range
Lin, H.Y., Hsieh, T.J., Tu, Z., Huang, B.S., Wu, S.C., Chien, C.T., Hsu, S.T., Lin, C.H.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Galectin-1 | A [auth B], B [auth A] | 154 | Homo sapiens | Mutation(s): 0 Gene Names: LGALS1 | 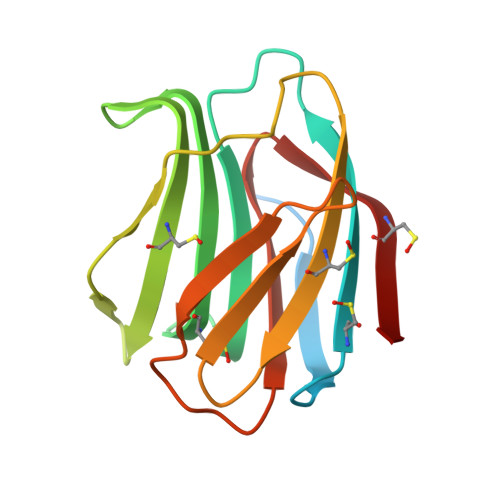 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P09382 GTEx: ENSG00000100097 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P09382 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Modified Residues 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| CSO Query on CSO | A [auth B], B [auth A] | L-PEPTIDE LINKING | C3 H7 N O3 S |  | CYS |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 43.3 | α = 90 |
| b = 58.234 | β = 90 |
| c = 111.649 | γ = 90 |
| Software Name | Purpose |
|---|---|
| Blu-Ice | data collection |
| HKL-2000 | data processing |
| HKL-2000 | data scaling |
| PHASER | phasing |
| PHENIX | refinement |
| PDB_EXTRACT | data extraction |
| Funding Organization | Location | Grant Number |
|---|---|---|
| Academia Sinica | Taiwan | AS-022316 |
| Ministry of Science and Technology | Taiwan | 102-2923-M-001-001-MY3 |
| Ministry of Science and Technology | Taiwan | 102-2113-M-001-001-MY3 |
| Ministry of Science and Technology | Taiwan | 103-2113-M-001-023-MY3 |