Crystal structure of yeast N-terminal acetyltransferase NatE (IP6) in complex with a bisubstrate
Dong, J., Wang, S., York, J.D.To be published.
Experimental Data Snapshot
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| N-terminal acetyltransferase A complex subunit NAT1 | 854 | Saccharomyces cerevisiae | Mutation(s): 0 | 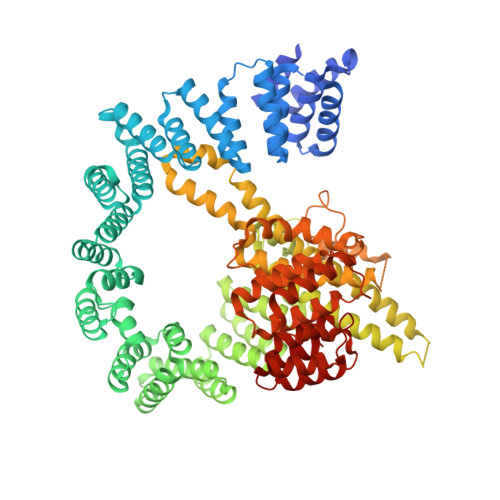 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P12945 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| N-terminal acetyltransferase A complex catalytic subunit ARD1 | 238 | Saccharomyces cerevisiae | Mutation(s): 0 EC: 2.3.1.88 (PDB Primary Data), 2.3.1.255 (UniProt) | 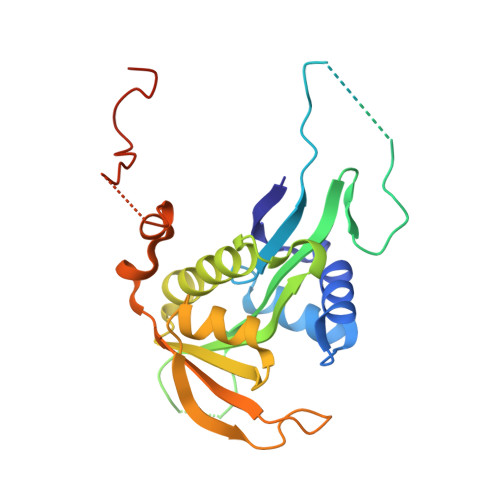 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P07347 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| N-terminal acetyltransferase A complex subunit NAT5 | 176 | Saccharomyces cerevisiae | Mutation(s): 0 EC: 2.3.1 (PDB Primary Data), 2.3.1.258 (UniProt) | 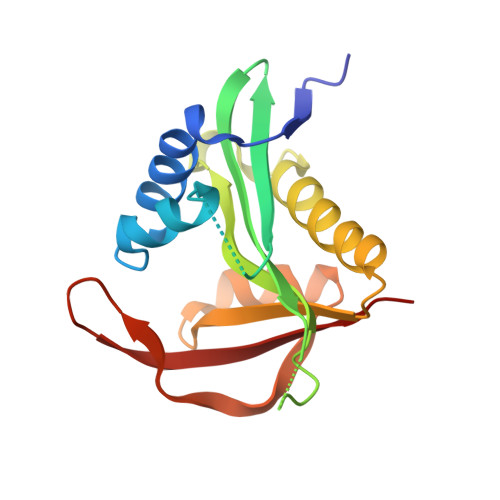 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q08689 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 4 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| ACYH8 | D [auth F] | 8 | Homo sapiens | Mutation(s): 0 | 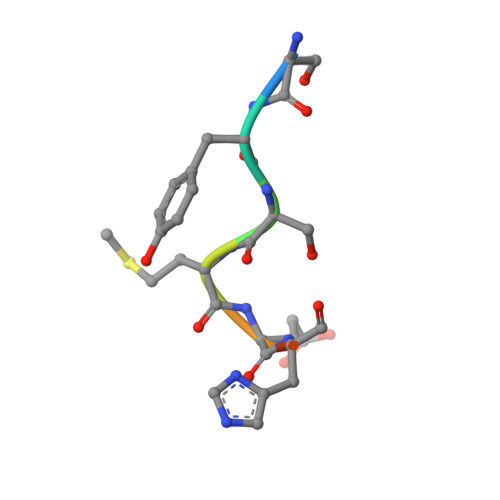 |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P01189 GTEx: ENSG00000115138 | |||||
Entity Groups | |||||
| UniProt Group | P01189 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| CMC Download:Ideal Coordinates CCD File | G [auth F] | CARBOXYMETHYL COENZYME *A C23 H38 N7 O18 P3 S OBUOSIHPWVNVJN-GRFIIANRSA-N |  | ||
| ACO Download:Ideal Coordinates CCD File | F [auth C] | ACETYL COENZYME *A C23 H38 N7 O17 P3 S ZSLZBFCDCINBPY-ZSJPKINUSA-N |  | ||
| IHP Download:Ideal Coordinates CCD File | E [auth A] | INOSITOL HEXAKISPHOSPHATE C6 H18 O24 P6 IMQLKJBTEOYOSI-GPIVLXJGSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 84.475 | α = 90 |
| b = 114.515 | β = 90 |
| c = 146.978 | γ = 90 |
| Software Name | Purpose |
|---|---|
| PHENIX | refinement |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |
| PHASER | phasing |