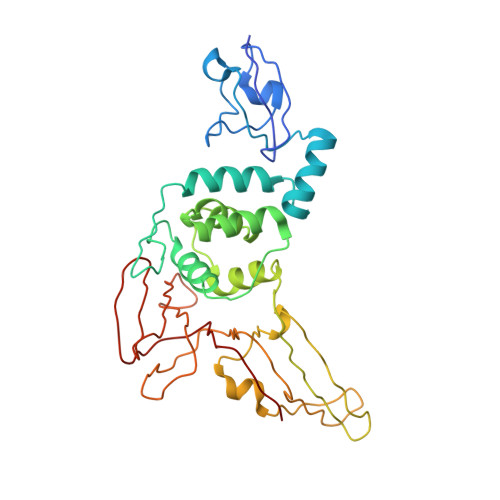
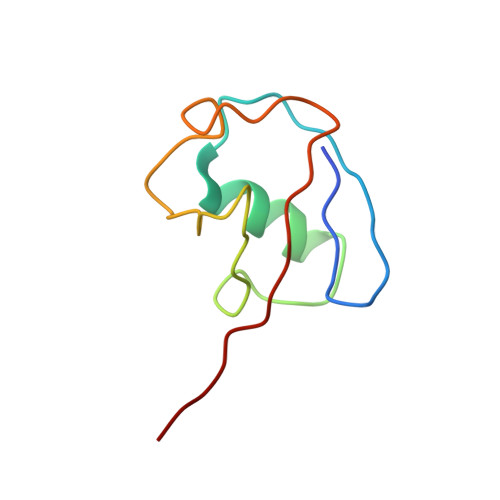
Structural and mutational analysis of the interaction between the Middle-East respiratory syndrome coronavirus (MERS-CoV) papain-like protease and human ubiquitin.
Lei, J., Hilgenfeld, R.(2016) Virol Sin 31: 288-299
- PubMed: 27245450 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1007/s12250-016-3742-4
- Primary Citation Related Structures:
4WUR, 5HIH - PubMed Abstract:
The papain-like protease (PL(pro)) of Middle-East respiratory syndrome coronavirus (MERS-CoV) has proteolytic, deubiquitinating, and deISGylating activities. The latter two are involved in the suppression of the antiviral innate immune response of the host cell. To contribute to an understanding of this process, we present here the X-ray crystal structure of a complex between MERS-CoV PL(pro) and human ubiquitin (Ub) that is devoid of any covalent linkage between the two proteins. Five regions of the PL(pro) bind to two areas of the Ub. The C-terminal five residues of Ub, RLRGG, are similar to the P5-P1 residues of the polyprotein substrates of the PL(pro) and are responsible for the major part of the interaction between the two macromolecules. Through sitedirected mutagenesis, we demonstrate that conserved Asp165 and non-conserved Asp164 are important for the catalytic activities of MERS-CoV PL(pro). The enzyme appears not to be optimized for catalytic efficiency; thus, replacement of Phe269 by Tyr leads to increased peptidolytic and deubiquitinating activities. Ubiquitin binding by MERS-CoV PL(pro) involves remarkable differences compared to the corresponding complex with SARS-CoV PL(pro). The structure and the mutational study help understand common and unique features of the deubiquitinating activity of MERS-CoV PL(pro).
- Institute of Biochemistry, Center for Structural and Cell Biology in Medicine, University of Lübeck, 23562, Lübeck, Germany.
Organizational Affiliation: