Structure of yeast SAGA DUBm with Sgf73 N59D mutant at 2.36 angstroms resolution
Wolberger, C., Yan, M.To be published.
Experimental Data Snapshot
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Ubiquitin carboxyl-terminal hydrolase 8 | A, E [auth D] | 476 | Saccharomyces cerevisiae S288C | Mutation(s): 0 Gene Names: UBP8, YMR223W, YM9959.05 EC: 3.4.19.12 | 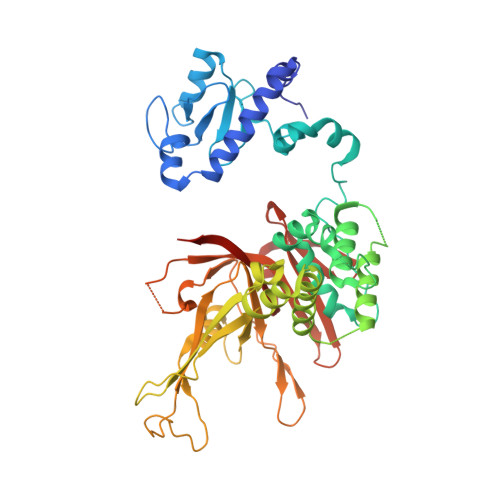 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P50102 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Transcription and mRNA export factor SUS1 | 96 | Saccharomyces cerevisiae S288C | Mutation(s): 0 Gene Names: SUS1, YBR111W-A |  | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q6WNK7 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 3 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| SAGA-associated factor 11 | 99 | Saccharomyces cerevisiae YJM789 | Mutation(s): 0 Gene Names: SGF11, SCY_5678 |  | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | A6ZWK1 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 4 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| SAGA-associated factor 73 | D [auth E], H | 96 | Saccharomyces cerevisiae S288C | Mutation(s): 1 Gene Names: SGF73, YGL066W | 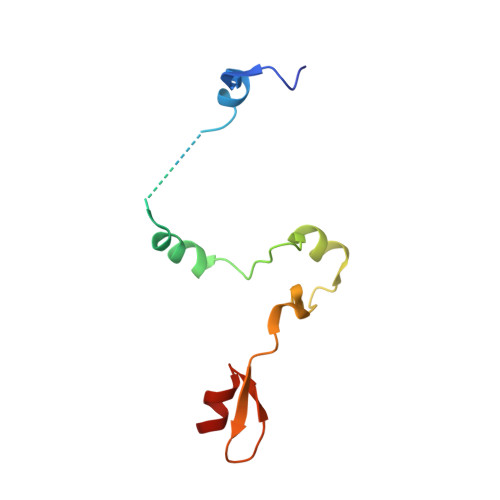 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P53165 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| ZN Download:Ideal Coordinates CCD File | I [auth A] J [auth A] K [auth A] L [auth A] M [auth A] | ZINC ION Zn PTFCDOFLOPIGGS-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 81.076 | α = 90 |
| b = 68.004 | β = 106.75 |
| c = 137.858 | γ = 90 |
| Software Name | Purpose |
|---|---|
| REFMAC | refinement |
| Blu-Ice | data collection |
| Coot | model building |
| HKL-2000 | data scaling |
| PHENIX | refinement |
| Funding Organization | Location | Grant Number |
|---|---|---|
| National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) | United States | GM-095822 |