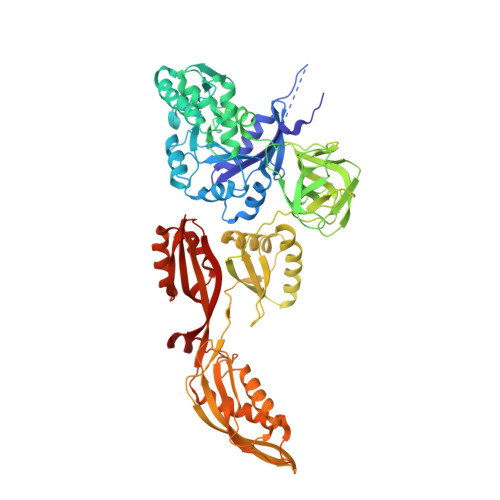
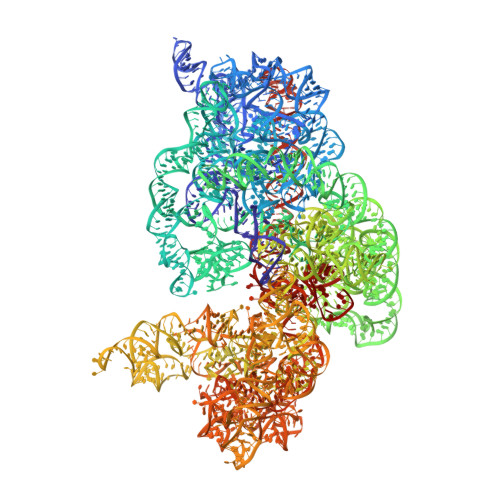
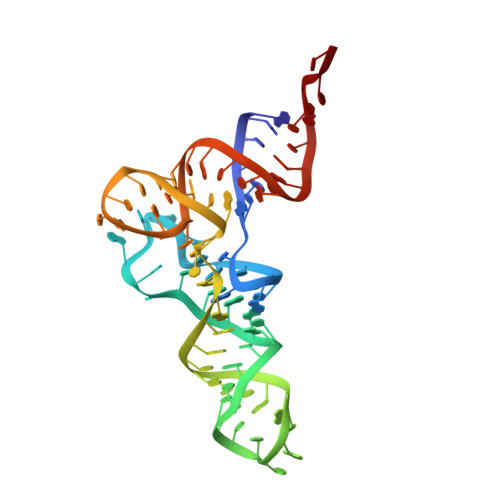
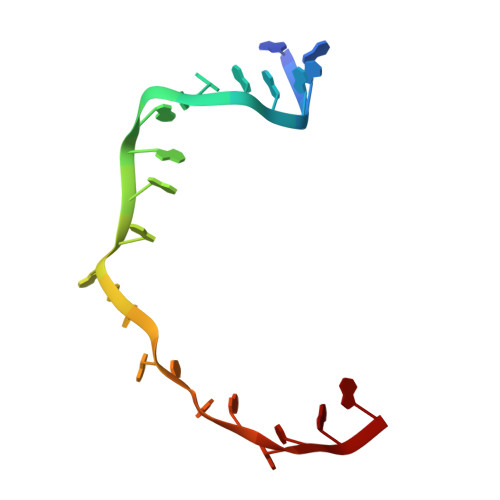
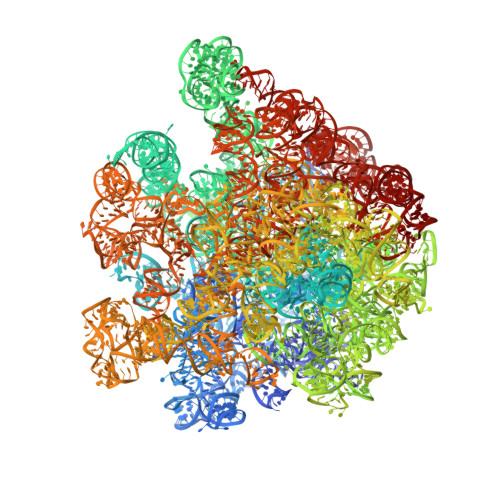
Crystal structures of EF-G-ribosome complexes trapped in intermediate states of translocation.
Zhou, J., Lancaster, L., Donohue, J.P., Noller, H.F.(2013) Science 340: 1236086-1236086
- PubMed: 23812722 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1126/science.1236086
- Primary Citation Related Structures:
4V9J, 4V9K, 4V9L, 4V9M - PubMed Abstract:
Translocation of messenger and transfer RNA (mRNA and tRNA) through the ribosome is a crucial step in protein synthesis, whose mechanism is not yet understood. The crystal structures of three Thermus ribosome-tRNA-mRNA-EF-G complexes trapped with β,γ-imidoguanosine 5'-triphosphate (GDPNP) or fusidic acid reveal conformational changes occurring during intermediate states of translocation, including large-scale rotation of the 30S subunit head and body. In all complexes, the tRNA acceptor ends occupy the 50S subunit E site, while their anticodon stem loops move with the head of the 30S subunit to positions between the P and E sites, forming chimeric intermediate states. Two universally conserved bases of 16S ribosomal RNA that intercalate between bases of the mRNA may act as "pawls" of a translocational ratchet. These findings provide new insights into the molecular mechanism of ribosomal translocation.
- Center for Molecular Biology of RNA and Department of Molecular, Cell and Developmental Biology, University of California, Santa Cruz, CA 95064, USA.
Organizational Affiliation: