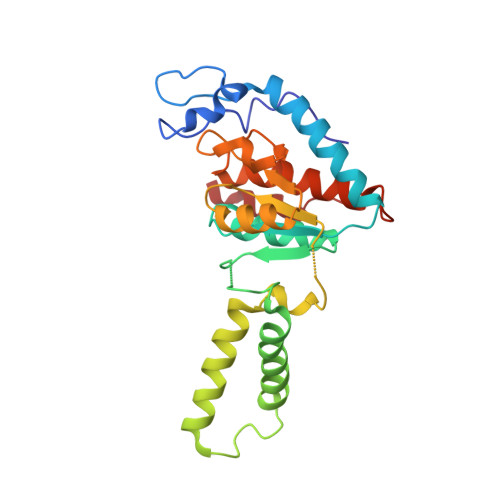
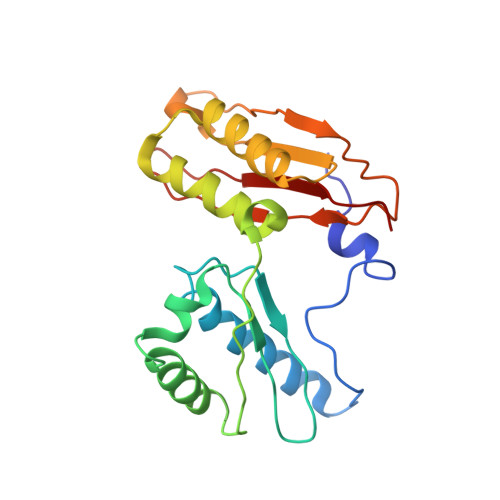
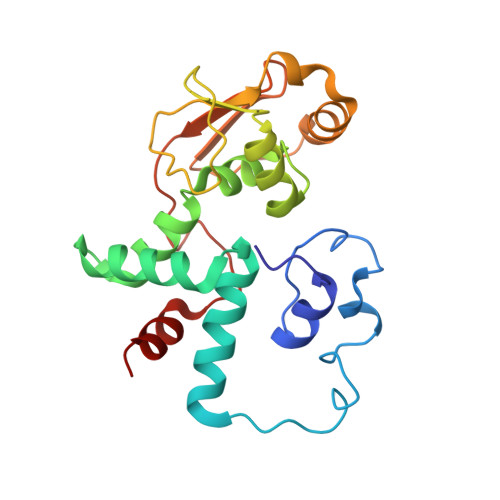
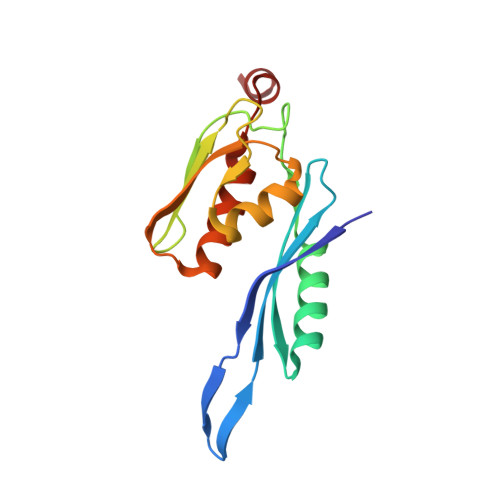
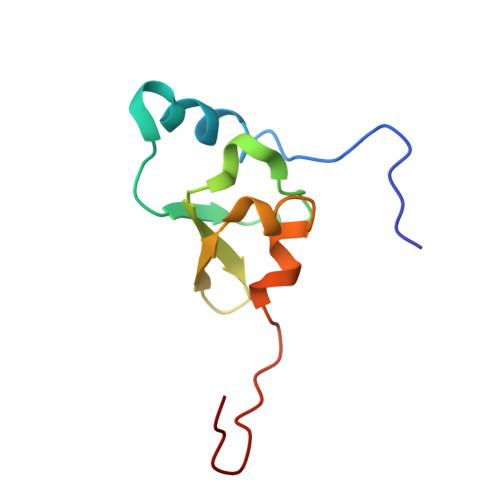
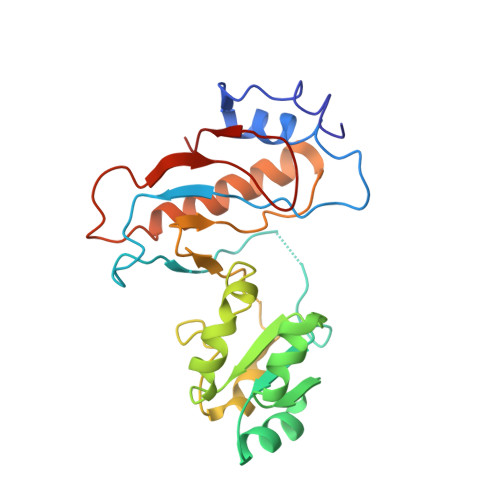
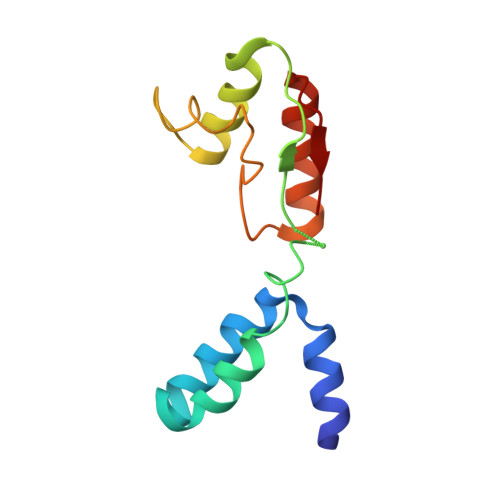
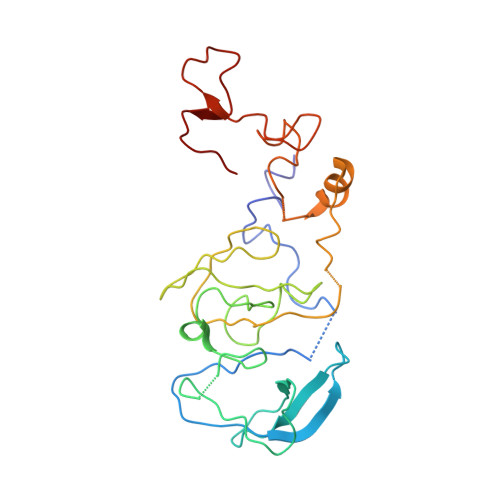
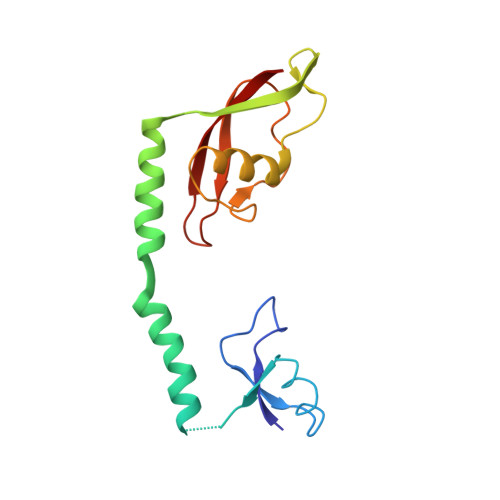
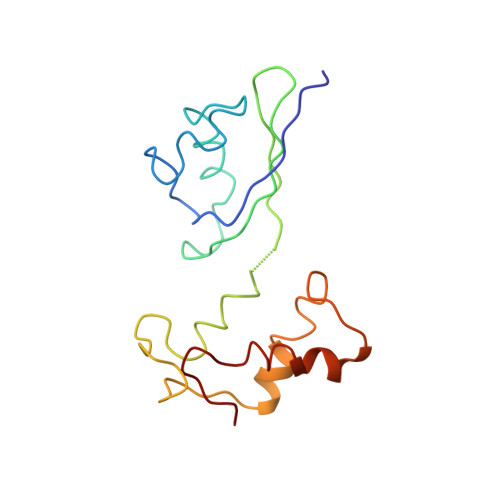
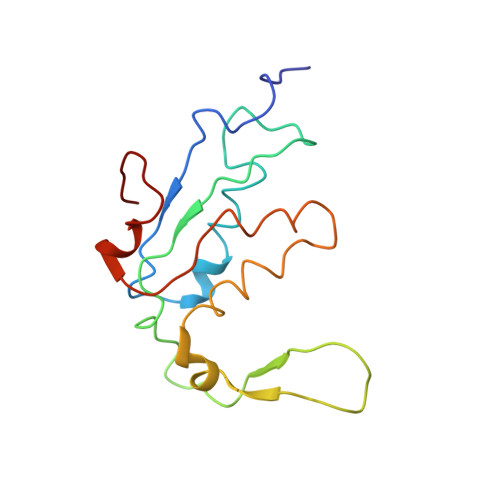
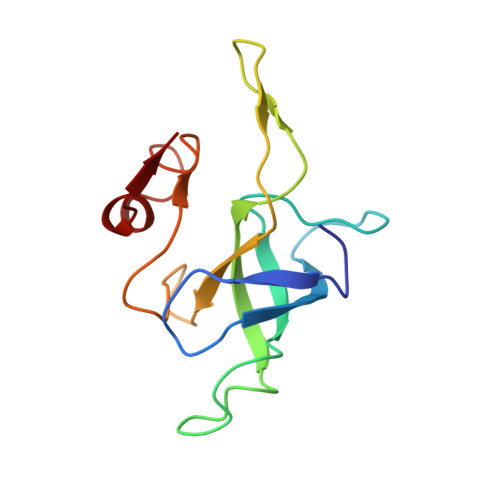
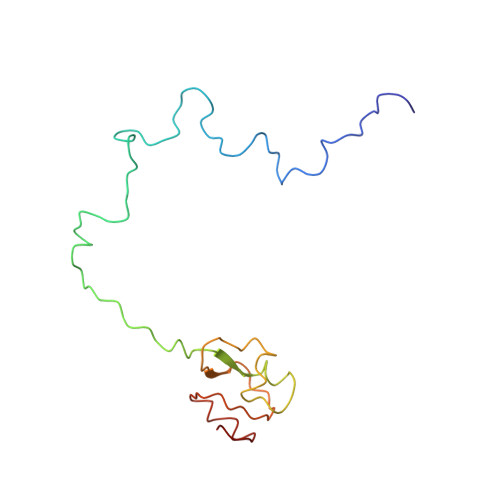
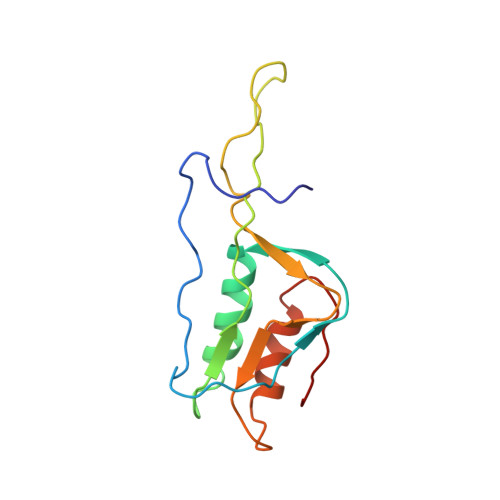
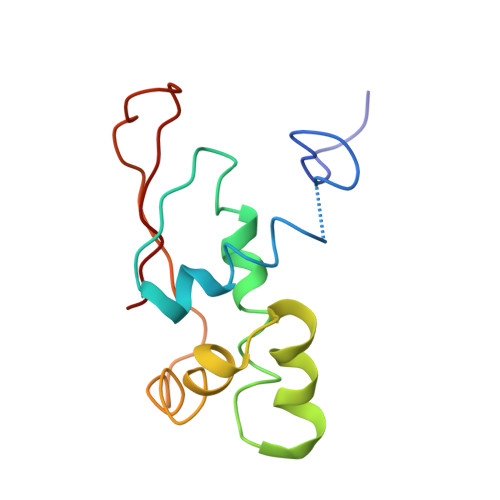
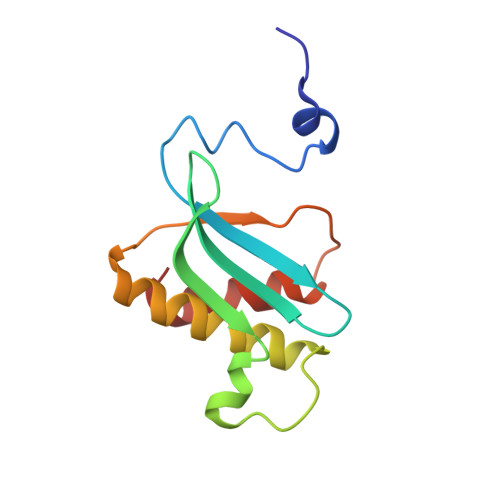
Elongation arrest by SecM via a cascade of ribosomal RNA rearrangements
Mitra, K., Schaffitzel, C., Fabiola, F., Chapman, M.S., Ban, N., Frank, J.(2006) Mol Cell 22: 533-543
- PubMed: 16713583 Search on PubMed
- DOI: https://doi.org/10.1016/j.molcel.2006.05.003
- Primary Citation Related Structures:
4V4V, 4V4W - PubMed Abstract:
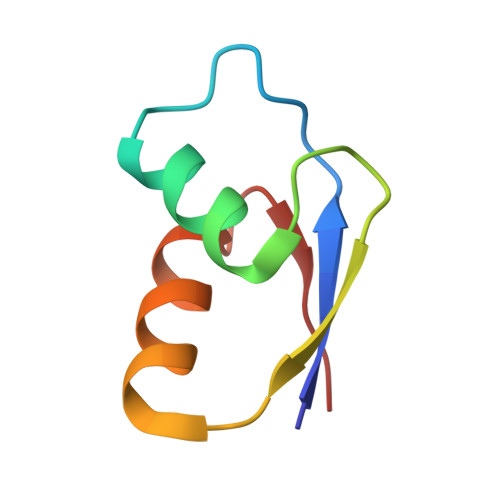
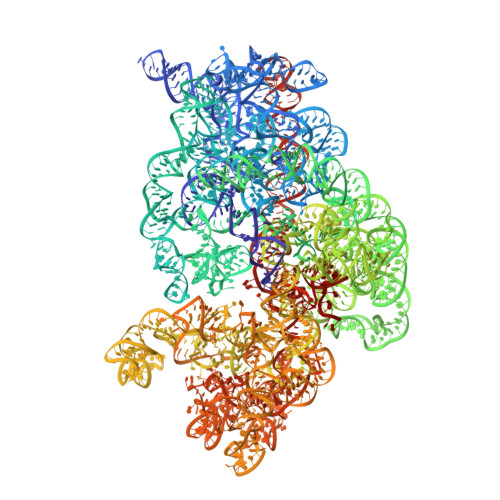
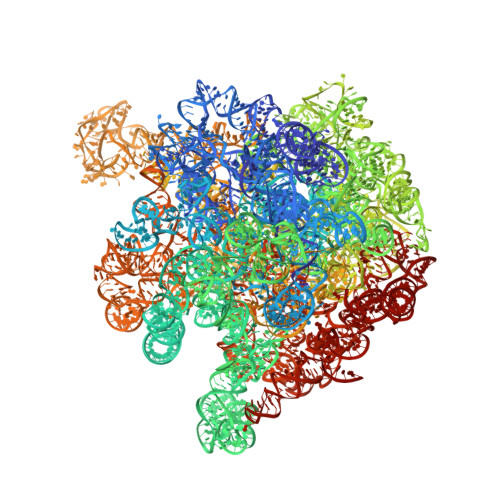
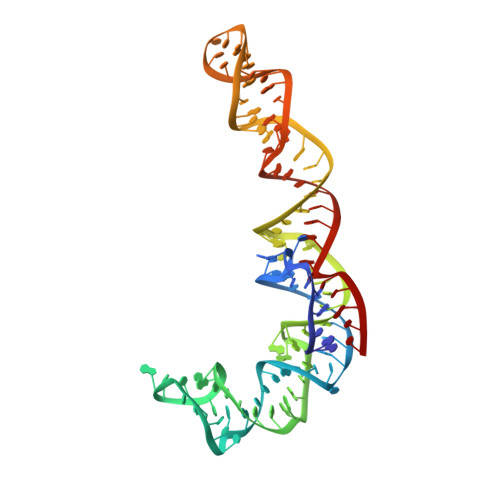
In E. coli, the SecM nascent polypeptide causes elongation arrest, while interacting with 23S RNA bases A2058 and A749-753 in the exit tunnel of the large ribosomal subunit. We compared atomic models fitted by real-space refinement into cryo-electron microscopy reconstructions of a pretranslocational and SecM-stalled E. coli ribosome complex. A cascade of RNA rearrangements propagates from the exit tunnel throughout the large subunit, affecting intersubunit bridges and tRNA positions, which in turn reorient small subunit RNA elements. Elongation arrest could result from the inhibition of mRNA.(tRNAs) translocation, E site tRNA egress, and perhaps translation factor activation at the GTPase-associated center. Our study suggests that the specific secondary and tertiary arrangement of ribosomal RNA provides the basis for internal signal transduction within the ribosome. Thus, the ribosome may itself have the ability to regulate its progression through translation by modulating its structure and consequently its receptivity to activation by cofactors.
- Howard Hughes Medical Institute, Health Research, Inc., at the Wadsworth Center, Empire State Plaza, Albany, New York 12201, USA.
Organizational Affiliation: