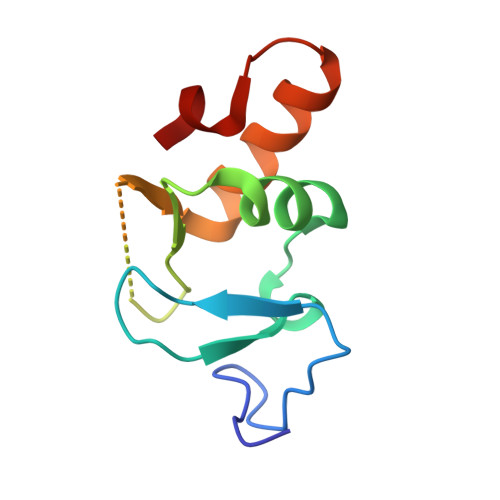
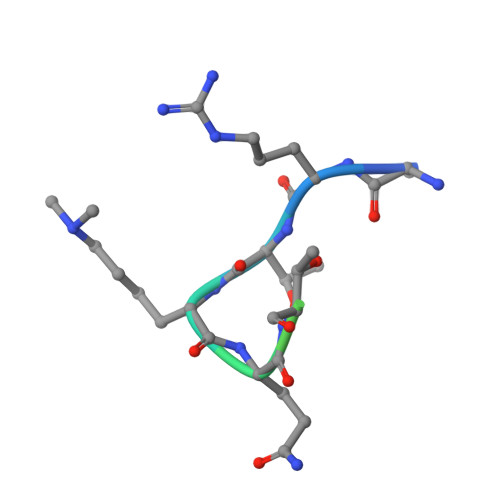
Competitive Binding of a Benzimidazole to the Histone-Binding Pocket of the Pygo Phd Finger.
Miller, T.C.R., Rutherford, T.J., Birchall, K., Chugh, J., Fiedler, M., Bienz, M.(2014) ACS Chem Biol 9: 2864
- PubMed: 25323450 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1021/cb500585s
- Primary Citation Related Structures:
4UP0, 4UP5 - PubMed Abstract:
The Pygo-BCL9 complex is a chromatin reader, facilitating β-catenin-mediated oncogenesis, and is thus emerging as a potential therapeutic target for cancer. Its function relies on two ligand-binding surfaces of Pygo's PHD finger that anchor the histone H3 tail methylated at lysine 4 (H3K4me) with assistance from the BCL9 HD1 domain. Here, we report the first use of fragment-based screening by NMR to identify small molecules that block protein-protein interactions by a PHD finger. This led to the discovery of a set of benzothiazoles that bind to a cleft emanating from the PHD-HD1 interface, as defined by X-ray crystallography. Furthermore, we discovered a benzimidazole that docks into the H3K4me specificity pocket and displaces the native H3K4me peptide from the PHD finger. Our study demonstrates the ligandability of the Pygo-BCL9 complex and uncovers a privileged scaffold as a template for future development of lead inhibitors of oncogenesis.
- MRC Laboratory of Molecular Biology , Francis Crick Avenue, Cambridge CB2 0QH, United Kingdom.
Organizational Affiliation: