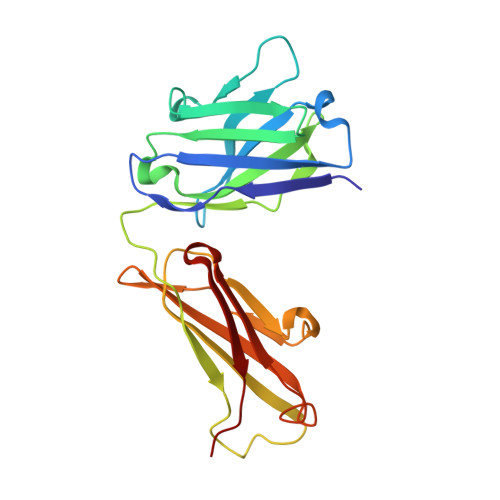
Locking and Blocking the Viral Landscape of an Alphavirus with Neutralizing Antibodies.
Porta, J., Jose, J., Roehrig, J.T., Blair, C.D., Kuhn, R.J., Rossmann, M.G.(2014) J Virol 88: 9616
- PubMed: 24920796 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1128/JVI.01286-14
- Primary Citation Related Structures:
4UOK, 4UOM - PubMed Abstract:
Alphaviruses are serious, sometimes lethal human pathogens that belong to the family Togaviridae. The structures of human Venezuelan equine encephalitis virus (VEEV), an alphavirus, in complex with two strongly neutralizing antibody Fab fragments (F5 and 3B4C-4) have been determined using a combination of cryo-electron microscopy and homology modeling. We characterize these monoclonal antibody Fab fragments, which are known to abrogate VEEV infectivity by binding to the E2 (envelope) surface glycoprotein. Both of these antibody Fab fragments cross-link the surface E2 glycoproteins and therefore probably inhibit infectivity by blocking the conformational changes that are required for making the virus fusogenic. The F5 Fab fragment cross-links E2 proteins within one trimeric spike, whereas the 3B4C-4 Fab fragment cross-links E2 proteins from neighboring spikes. Furthermore, F5 probably blocks the receptor-binding site, whereas 3B4C-4 sterically hinders the exposure of the fusion loop at the end of the E2 B-domain. Alphaviral infections are transmitted mainly by mosquitoes. Venezuelan equine encephalitis virus (VEEV) is an alphavirus with a wide distribution across the globe. No effective vaccines exist for alphaviral infections. Therefore, a better understanding of VEEV and its associated neutralizing antibodies will help with the development of effective drugs and vaccines.
- Department of Biological Sciences, Purdue University, West Lafayette, Indiana, USA.
Organizational Affiliation: