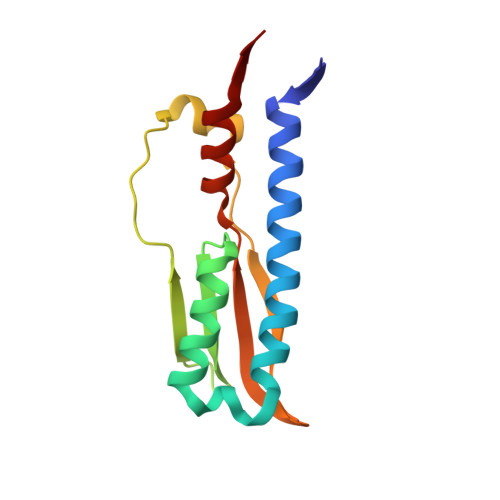
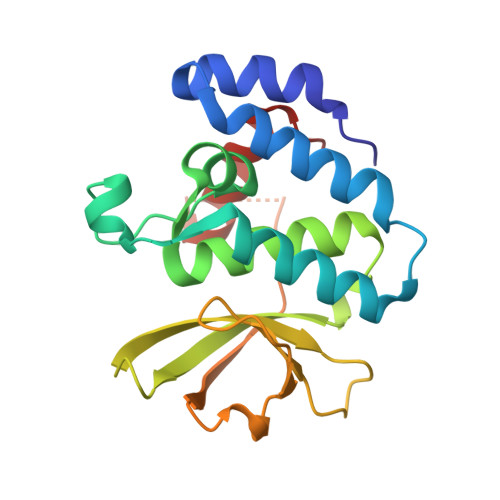
Mechanistic insight into the conserved allosteric regulation of periplasmic proteolysis by the signaling molecule cyclic-di-GMP.
Chatterjee, D., Cooley, R.B., Boyd, C.D., Mehl, R.A., O'Toole, G.A., Sondermann, H.(2014) Elife 3: e03650-e03650
- PubMed: 25182848 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.7554/eLife.03650
- Primary Citation Related Structures:
4U64, 4U65 - PubMed Abstract:
Stable surface adhesion of cells is one of the early pivotal steps in bacterial biofilm formation, a prevalent adaptation strategy in response to changing environments. In Pseudomonas fluorescens, this process is regulated by the Lap system and the second messenger cyclic-di-GMP. High cytoplasmic levels of cyclic-di-GMP activate the transmembrane receptor LapD that in turn recruits the periplasmic protease LapG, preventing it from cleaving a cell surface-bound adhesin, thereby promoting cell adhesion. In this study, we elucidate the molecular basis of LapG regulation by LapD and reveal a remarkably sensitive switching mechanism that is controlled by LapD's HAMP domain. LapD appears to act as a coincidence detector, whereby a weak interaction of LapG with LapD transmits a transient outside-in signal that is reinforced only when cyclic-di-GMP levels increase. Given the conservation of key elements of this receptor system in many bacterial species, the results are broadly relevant for cyclic-di-GMP- and HAMP domain-regulated transmembrane signaling.
- Department of Molecular Medicine, College of Veterinary Medicine, Cornell University, Ithaca, United States.
Organizational Affiliation: