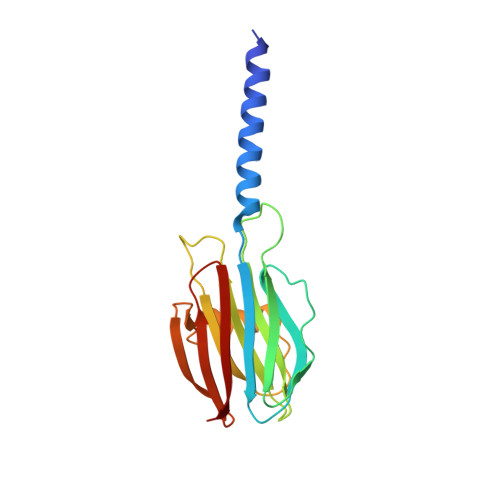
Structural basis for self-assembly of a cytolytic pore lined by protein and lipid
Tanaka, K., Caaveiro, J.M.M., Morante, K., Gonzalez-Manas, J.M., Tsumoto, K.(2015) Nat Commun 6: 6337-6337
- PubMed: 25716479 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/ncomms7337
- Primary Citation Related Structures:
3VWI, 3W9P, 4TSL, 4TSN, 4TSO, 4TSP, 4TSQ, 4TSY - PubMed Abstract:
Pore-forming toxins (PFT) are water-soluble proteins that possess the remarkable ability to self-assemble on the membrane of target cells, where they form pores causing cell damage. Here, we elucidate the mechanism of action of the haemolytic protein fragaceatoxin C (FraC), a α-barrel PFT, by determining the crystal structures of FraC at four different stages of the lytic mechanism, namely the water-soluble state, the monomeric lipid-bound form, an assembly intermediate and the fully assembled transmembrane pore. The structure of the transmembrane pore exhibits a unique architecture composed of both protein and lipids, with some of the lipids lining the pore wall, acting as assembly cofactors. The pore also exhibits lateral fenestrations that expose the hydrophobic core of the membrane to the aqueous environment. The incorporation of lipids from the target membrane within the structure of the pore provides a membrane-specific trigger for the activation of a haemolytic toxin.
- Department of Chemistry and Biotechnology, School of Engineering, The University of Tokyo, Bunkyo-ku, Tokyo 113-8656, Japan.
Organizational Affiliation: