Elucidation of the mechanism of disulfide exchange between staphylococcal thioredoxin2 and thioredoxin reductase2: A structural insight.
Bose, M., Bhattacharyya, S., Biswas, R., Roychowdhury, A., Bhattacharjee, A., Ghosh, A.K., Das, A.K.(2019) Biochimie 160: 1-13
- PubMed: 30710560 Search on PubMed
- DOI: https://doi.org/10.1016/j.biochi.2019.01.019
- Primary Citation Related Structures:
4RUV, 6A4J - PubMed Abstract:
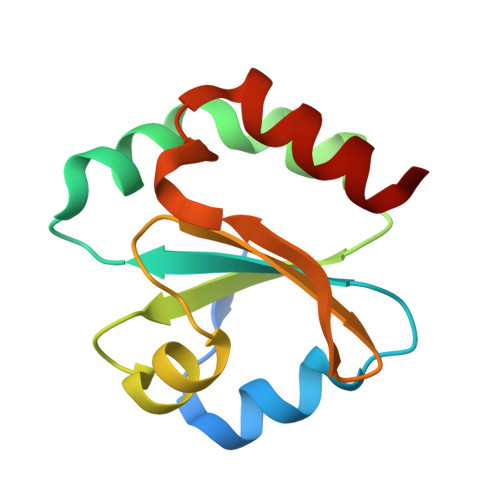
The redox homeostasis of cytoplasm is maintained by a series of disulfide exchange reactions mediated by proteins belonging to the thioredoxin superfamily. Thioredoxin and thioredoxin reductase, being the major members of the family, play a key role in oxidative stress response of Staphylococcus aureus. In this report, we have identified and characterised an active thioredoxin system of the mentioned pathogen. Crystal structure of thioredoxin2 (SaTrx2) in its reduced form reveals that it contains the conserved redox active WCXXC motif and a thioredoxin fold. Thioredoxin reductase2 (SaTR2) is a flavoprotein and consists of two Rossmann folds as the binding sites for FAD and NADPH. Crystal structure of the SaTR2 holoenzyme shows that the protein consists of two domains and the catalytic site comprises of an intramolecular disulfide bond formed between two sequentially distal cysteine residues. Biophysical and biochemical studies unveil that SaTrx2 and SaTR2 can physically interact in solution and in the course of sustaining the redox equilibrium, the latter reduces the former. Molecular docking has been performed to illustrate the interface formed between SaTrx2 and SaTR2 during the disulfide exchange reaction.
- Department of Biotechnology, Indian Institute of Technology Kharagpur, Kharagpur, 721302, India.
Organizational Affiliation: