Crystal Structure of Human Presequence Protease in Complex with Inhibitor MitoBloCK-60
Mo, S.M., Liang, W.G., King, J.V., Wijaya, J., Koehler, C.M., Tang, W.J.To be published.
Experimental Data Snapshot
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Presequence protease, mitochondrial | 1,014 | Homo sapiens | Mutation(s): 1 Gene Names: PITRM1, KIAA1104, MP1 EC: 3.4.24 | 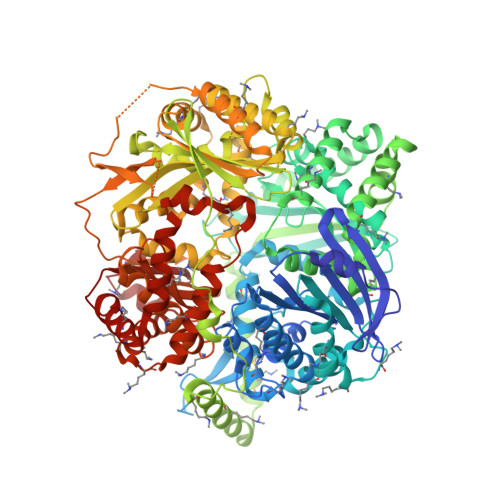 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q5JRX3 GTEx: ENSG00000107959 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q5JRX3 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Presequence protease, mitochondrial | 1,014 | Homo sapiens | Mutation(s): 1 Gene Names: PITRM1, KIAA1104, MP1 EC: 3.4.24 | 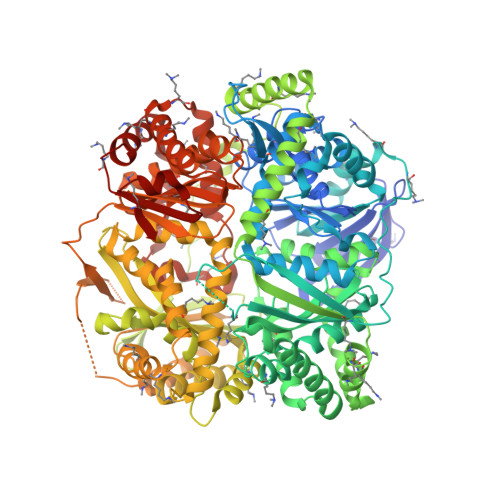 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: Q5JRX3 GTEx: ENSG00000107959 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q5JRX3 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 5 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| 3UE Download:Ideal Coordinates CCD File | D [auth A], E [auth A], U [auth B], V [auth B] | [4-(diphenylmethyl)piperazin-1-yl](3-methyl-4-nitrophenyl)methanone C25 H25 N3 O3 FJZPDGSOVGNVJO-UHFFFAOYSA-N |  | ||
| GOL Download:Ideal Coordinates CCD File | AA [auth B] BA [auth B] CA [auth B] F [auth A] G [auth A] | GLYCEROL C3 H8 O3 PEDCQBHIVMGVHV-UHFFFAOYSA-N |  | ||
| ZN Download:Ideal Coordinates CCD File | C [auth A], T [auth B] | ZINC ION Zn PTFCDOFLOPIGGS-UHFFFAOYSA-N |  | ||
| ACT Download:Ideal Coordinates CCD File | DA [auth B] EA [auth B] FA [auth B] GA [auth B] HA [auth B] | ACETATE ION C2 H3 O2 QTBSBXVTEAMEQO-UHFFFAOYSA-M |  | ||
| CA Download:Ideal Coordinates CCD File | IA [auth B], JA [auth B] | CALCIUM ION Ca BHPQYMZQTOCNFJ-UHFFFAOYSA-N |  | ||
| Modified Residues 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Type | Formula | 2D Diagram | Parent |
| CAS Query on CAS | A | L-PEPTIDE LINKING | C5 H12 As N O2 S |  | CYS |
| MLY Query on MLY | A | L-PEPTIDE LINKING | C8 H18 N2 O2 |  | LYS |
| MLZ Query on MLZ | A | L-PEPTIDE LINKING | C7 H16 N2 O2 |  | LYS |
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 245.624 | α = 90 |
| b = 85.49 | β = 127.53 |
| c = 158.214 | γ = 90 |
| Software Name | Purpose |
|---|---|
| HKL-3000 | data collection |
| PHASER | phasing |
| PHENIX | refinement |
| HKL-3000 | data reduction |
| HKL-3000 | data scaling |