Crystal structure of a distal-less homeobox protein 5 (Dlx5) from Homo sapiens at 1.85 A resolution
Joint Center for Structural Genomics (JCSG), Partnership for Stem Cell Biology (STEMCELL)To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Homeobox protein DLX-5 | 65 | Homo sapiens | Mutation(s): 2 Gene Names: BC006226, DLX5 | 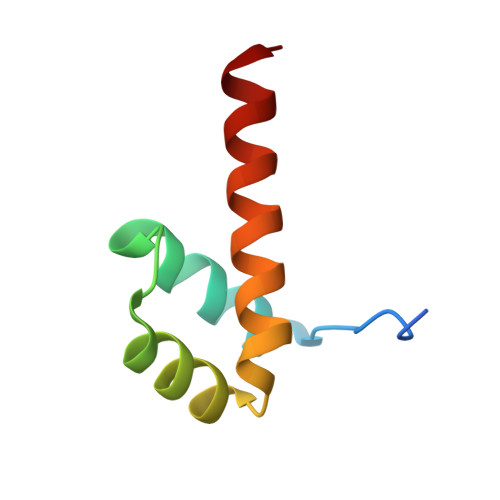 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P56178 GTEx: ENSG00000105880 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P56178 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 34.182 | α = 84.13 |
| b = 42.913 | β = 77.33 |
| c = 56.104 | γ = 67.13 |
| Software Name | Purpose |
|---|---|
| MolProbity | model building |
| PDB_EXTRACT | data extraction |
| PHASER | phasing |
| SCALA | data scaling |
| REFMAC | refinement |
| MOSFLM | data reduction |