Crystal structure of heme oxygenase 2 from Chlamydomonas reinhardtii without heme.
Lopez, O., Duanmu, D., Lagarias, J.C., Fisher, A.J.To be published.
Experimental Data Snapshot
Starting Models: experimental
View more details
wwPDB Validation 3D Report Full Report
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Heme oxygenase | 247 | Chlamydomonas reinhardtii | Mutation(s): 0 Gene Names: CHLREDRAFT_152591, HMOX2 EC: 1.14.99.3 | 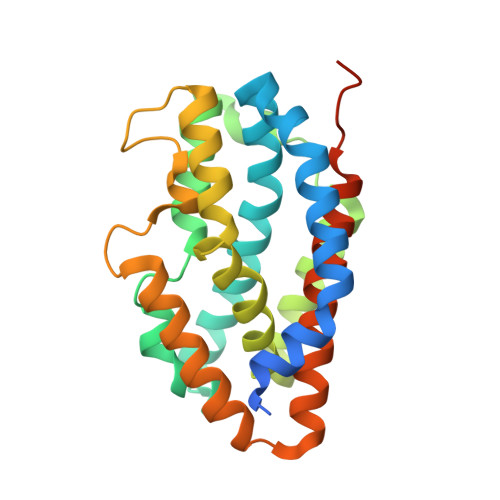 | |
| Ligands 1 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| PG6 Download:Ideal Coordinates CCD File | B [auth A] | 1-(2-METHOXY-ETHOXY)-2-{2-[2-(2-METHOXY-ETHOXY]-ETHOXY}-ETHANE C12 H26 O6 DMDPGPKXQDIQQG-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 41.93 | α = 90 |
| b = 134.77 | β = 90 |
| c = 80.69 | γ = 90 |
| Software Name | Purpose |
|---|---|
| Blu-Ice | data collection |
| PHASER | phasing |
| PHENIX | refinement |
| XDS | data reduction |
| XSCALE | data scaling |