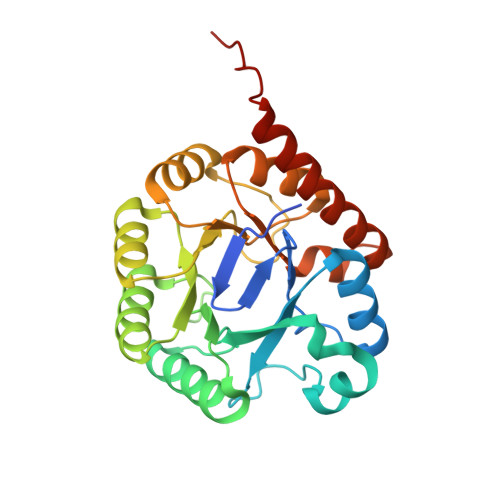
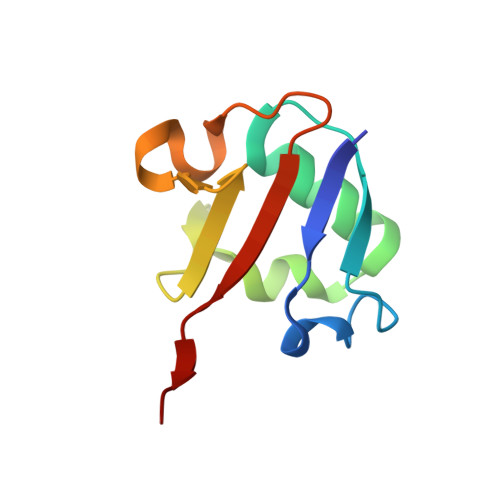
Co-opting sulphur-carrier proteins from primary metabolic pathways for 2-thiosugar biosynthesis.
Sasaki, E., Zhang, X., Sun, H.G., Lu, M.Y., Liu, T.L., Ou, A., Li, J.Y., Chen, Y.H., Ealick, S.E., Liu, H.W.(2014) Nature 509: 427-431
- PubMed: 24814342 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/nature13256
- Primary Citation Related Structures:
4N6E, 4N6F - PubMed Abstract:
Sulphur is an essential element for life and is ubiquitous in living systems. Yet how the sulphur atom is incorporated into many sulphur-containing secondary metabolites is poorly understood. For bond formation between carbon and sulphur in primary metabolites, the major ionic sulphur sources are the persulphide and thiocarboxylate groups on sulphur-carrier (donor) proteins. Each group is post-translationally generated through the action of a specific activating enzyme. In all reported bacterial cases, the gene encoding the enzyme that catalyses the carbon-sulphur bond formation reaction and that encoding the cognate sulphur-carrier protein exist in the same gene cluster. To study the production of the 2-thiosugar moiety in BE-7585A, an antibiotic from Amycolatopsis orientalis, we identified a putative 2-thioglucose synthase, BexX, whose protein sequence and mode of action seem similar to those of ThiG, the enzyme that catalyses thiazole formation in thiamine biosynthesis. However, no gene encoding a sulphur-carrier protein could be located in the BE-7585A cluster. Subsequent genome sequencing uncovered a few genes encoding sulphur-carrier proteins that are probably involved in the biosynthesis of primary metabolites but only one activating enzyme gene in the A. orientalis genome. Further experiments showed that this activating enzyme can adenylate each of these sulphur-carrier proteins and probably also catalyses the subsequent thiolation, through its rhodanese domain. A proper combination of these sulphur-delivery systems is effective for BexX-catalysed 2-thioglucose production. The ability of BexX to selectively distinguish sulphur-carrier proteins is given a structural basis using X-ray crystallography. This study is, to our knowledge, the first complete characterization of thiosugar formation in nature and also demonstrates the receptor promiscuity of the A. orientalis sulphur-delivery system. Our results also show that co-opting the sulphur-delivery machinery of primary metabolism for the biosynthesis of sulphur-containing natural products is probably a general strategy found in nature.
- Department of Chemistry, University of Texas at Austin, Austin, Texas 78712, USA.
Organizational Affiliation: