Structural insight into BL-induced activation of the BRI1-BAK1 complex
Sun, Y., Han, Z., Chai, J.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Protein BRASSINOSTEROID INSENSITIVE 1 | 767 | Arabidopsis thaliana | Mutation(s): 0 Gene Names: BRI1, At4g39400, F23K16.30 EC: 2.7.10.1 (PDB Primary Data), 2.7.11.1 (PDB Primary Data) | 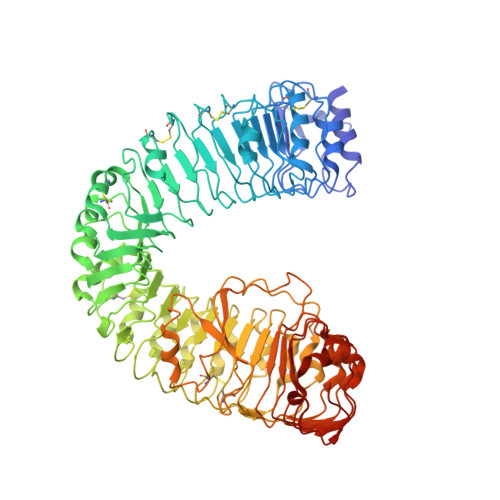 | |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | O22476 | ||||
Glycosylation | |||||
| Glycosylation Sites: 3 | |||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
Entity ID: 2 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| BRASSINOSTEROID INSENSITIVE 1-associated receptor kinase 1 | C [auth D], D [auth C] | 201 | Arabidopsis thaliana | Mutation(s): 0 Gene Names: BAK1, ELG, SERK3, At4g33430, F17M5.190 EC: 2.7.10.1 (PDB Primary Data), 2.7.11.1 (PDB Primary Data) | 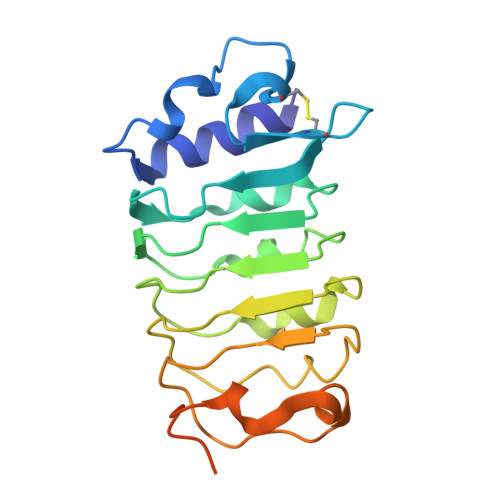 |
UniProt | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | Q94F62 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| BLD Download:Ideal Coordinates CCD File | L [auth A], O [auth B] | Brassinolide C28 H48 O6 IXVMHGVQKLDRKH-KNBKMWSGSA-N |  | ||
| NAG Download:Ideal Coordinates CCD File | I [auth A], J [auth A], M [auth B] | 2-acetamido-2-deoxy-beta-D-glucopyranose C8 H15 N O6 OVRNDRQMDRJTHS-FMDGEEDCSA-N |  | ||
| SO4 Download:Ideal Coordinates CCD File | K [auth A], N [auth B] | SULFATE ION O4 S QAOWNCQODCNURD-UHFFFAOYSA-L |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 145.911 | α = 90 |
| b = 145.911 | β = 90 |
| c = 166.24 | γ = 120 |
| Software Name | Purpose |
|---|---|
| HKL-2000 | data collection |
| PHASES | phasing |
| PHENIX | refinement |
| HKL-2000 | data reduction |
| HKL-2000 | data scaling |