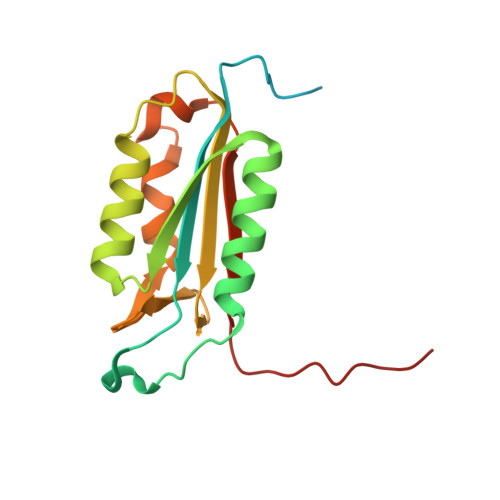
Combined inhibition of caspase 3 and caspase 7 by two highly selective DARPins slows down cellular demise.
Flutsch, A., Ackermann, R., Schroeder, T., Lukarska, M., Hausammann, G.J., Weinert, C., Briand, C., Grutter, M.G.(2014) Biochem J 461: 279-290
- PubMed: 24779913 Search on PubMed
- DOI: https://doi.org/10.1042/BJ20131456
- Primary Citation Related Structures:
4LSZ - PubMed Abstract:
Caspases play important roles during apoptosis, inflammation and proliferation. The high homology among family members makes selective targeting of individual caspases difficult, which is necessary to precisely define the role of these enzymes. We have selected caspase-7-specific binders from a library of DARPins (designed ankyrin repeat proteins). The DARPins D7.18 and D7.43 bind specifically to procaspase 7 and active caspase 7, but not to other members of the family. Binding of the DARPins does not affect the active enzyme, but interferes with its activation by other caspases. The crystal structure of the caspase 7-D7.18 complex elucidates the high selectivity and the mode of inhibition. Combining these caspase-7-specific DARPins with the previously reported caspase-3-inhibitory DARPin D3.4S76R reduces the activity of caspase 3 and 7 in double-transfected HeLa cells during apoptosis. In addition, these cells showed less susceptibility to TRAIL (tumour-necrosis-factor-related apoptosis-inducing ligand)-induced apoptosis in living cell experiments. D7.18 and D7.43 are therefore novel tools for in vitro studies on procaspase 7 activation as well as for clarifying the role of its activation in different cellular processes. If applied in combination with D3.4S76R, they represent an excellent instrument to increase our understanding of these enzymes during various cellular processes.
- *Department of Biochemistry, University of Zurich, Winterthurerstrasse 190, CH-8057 Zurich, Switzerland.
Organizational Affiliation: