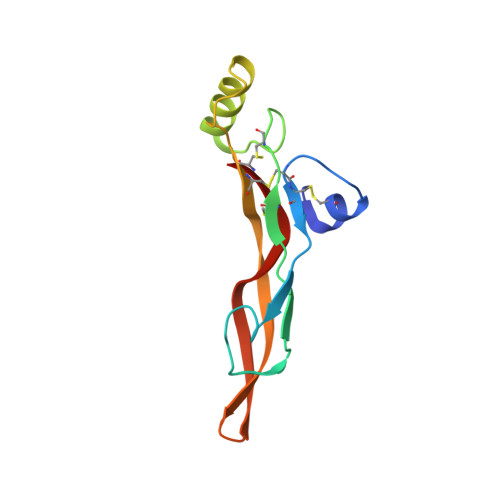
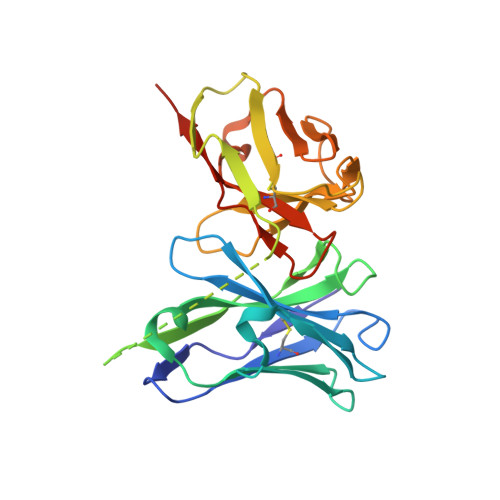
Structures of a pan-specific antagonist antibody complexed to different isoforms of TGF beta reveal structural plasticity of antibody-antigen interactions.
Moulin, A., Mathieu, M., Lawrence, C., Bigelow, R., Levine, M., Hamel, C., Marquette, J.P., Le Parc, J., Loux, C., Ferrari, P., Capdevila, C., Dumas, J., Dumas, B., Rak, A., Bird, J., Qiu, H., Pan, C.Q., Edmunds, T., Wei, R.R.(2014) Protein Sci 23: 1698-1707
- PubMed: 25209176 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1002/pro.2548
- Primary Citation Related Structures:
4KV5, 4KXZ - PubMed Abstract:
Various important biological pathways are modulated by TGFβ isoforms; as such they are potential targets for therapeutic intervention. Fresolimumab, also known as GC1008, is a pan-TGFβ neutralizing antibody that has been tested clinically for several indications including an ongoing trial for focal segmental glomerulosclerosis. The structure of the antigen-binding fragment of fresolimumab (GC1008 Fab) in complex with TGFβ3 has been reported previously, but the structural capacity of fresolimumab to accommodate tight interactions with TGFβ1 and TGFβ2 was insufficiently understood. We report the crystal structure of the single-chain variable fragment of fresolimumab (GC1008 scFv) in complex with target TGFβ1 to a resolution of 3.00 Å and the crystal structure of GC1008 Fab in complex with TGFβ2 to 2.83 Å. The structures provide further insight into the details of TGFβ recognition by fresolimumab, give a clear indication of the determinants of fresolimumab pan-specificity and provide potential starting points for the development of isoform-specific antibodies using a fresolimumab scaffold.
- Sanofi Biotherapeutics, Framingham, Massachusetts, USA.
Organizational Affiliation: