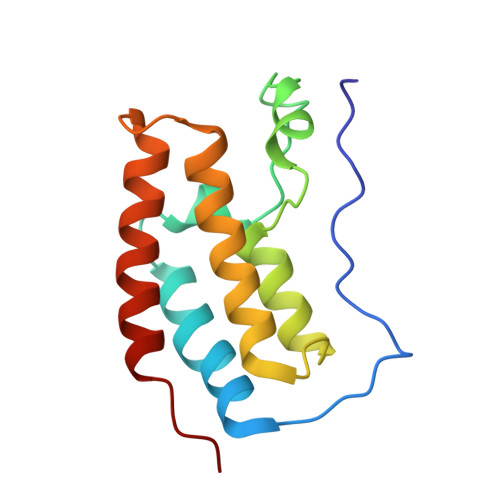
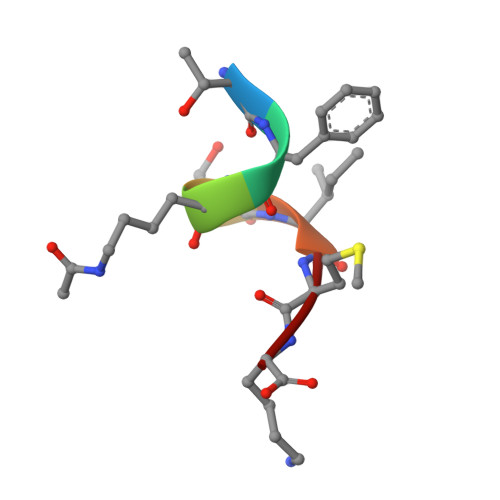
Brd4 maintains constitutively active NF-kappa B in cancer cells by binding to acetylated RelA.
Zou, Z., Huang, B., Wu, X., Zhang, H., Qi, J., Bradner, J., Nair, S., Chen, L.F.(2014) Oncogene 33: 2395-2404
- PubMed: 23686307 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1038/onc.2013.179
- Primary Citation Related Structures:
4KV1, 4KV4 - PubMed Abstract:
Acetylation of the RelA subunit of NF-κB at lysine-310 regulates the transcriptional activation of NF-κB target genes and contributes to maintaining constitutively active NF-κB in tumors. Bromodomain-containing factor Brd4 has been shown to bind to acetylated lysine-310 (AcLys310) and to regulate the transcriptional activity of NF-κB, but the role of this binding in maintaining constitutively active NF-κB in tumors remains elusive. In this study, we demonstrate the structural basis for the binding of bromodomains (BDs) of bromodomain-containing protein 4 (Brd4) to AcLys310 and identify the BD inhibitor JQ1 as an effective small molecule to block this interaction. JQ1 suppresses TNF-α-mediated NF-κB activation and NF-κB-dependent target gene expression. In addition, JQ1 inhibits the proliferation and transformation potential of A549 lung cancer cells and suppresses the tumorigenicity of A549 cells in severe combined immunodeficiency mice. Furthermore, we demonstrate that depletion of Brd4 or treatment of cells with JQ1 induces the ubiquitination and degradation of the constitutively active nuclear form of RelA. Our results identify a novel function of Brd4 in maintaining the persistently active form of NF-κB found in tumors, and they suggest that interference with the interaction between acetylated RelA and Brd4 could be a potential therapeutic approach for the treatment of NF-κB-driven cancer.
- Department of Biochemistry, College of Medicine, University of Illinois at Urbana-Champaign, Urbana, IL, USA.
Organizational Affiliation: