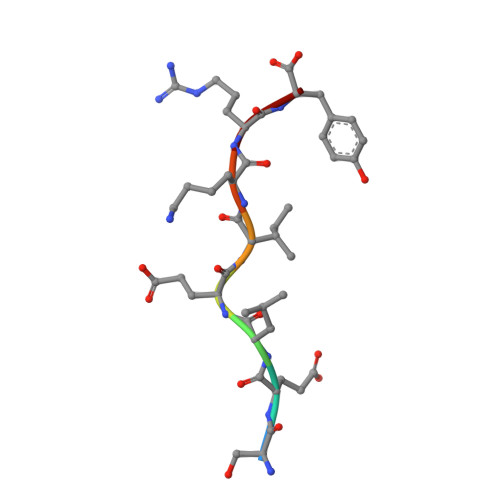
HLA Peptide Length Preferences Control CD8+ T Cell Responses.
Rist, M.J., Theodossis, A., Croft, N.P., Neller, M.A., Welland, A., Chen, Z., Sullivan, L.C., Burrows, J.M., Miles, J.J., Brennan, R.M., Gras, S., Khanna, R., Brooks, A.G., McCluskey, J., Purcell, A.W., Rossjohn, J., Burrows, S.R.(2013) J Immunol 191: 561-571
- PubMed: 23749632 Search on PubMed
- DOI: https://doi.org/10.4049/jimmunol.1300292
- Primary Citation Related Structures:
4JQV, 4JQX - PubMed Abstract:
Class I HLAs generally present peptides of 8-10 aa in length, although it is unclear whether peptide length preferences are affected by HLA polymorphism. In this study, we investigated the CD8(+) T cell response to the BZLF1 Ag of EBV, which includes overlapping sequences of different size that nevertheless conform to the binding motif of the large and abundant HLA-B*44 supertype. Whereas HLA-B*18:01(+) individuals responded strongly and exclusively to the octamer peptide (173)SELEIKRY(180), HLA-B*44:03(+) individuals responded to the atypically large dodecamer peptide (169)EECDSELEIKRY(180), which encompasses the octamer peptide. Moreover, the octamer peptide bound more stably to HLA-B*18:01 than did the dodecamer peptide, whereas, conversely, HLA-B*44:03 bound only the longer peptide. Furthermore, crystal structures of these viral peptide-HLA complexes showed that the Ag-binding cleft of HLA-B*18:01 was more ideally suited to bind shorter peptides, whereas HLA-B*44:03 exhibited characteristics that favored the presentation of longer peptides. Mass spectrometric identification of > 1000 naturally presented ligands revealed that HLA-B*18:01 was more biased toward presenting shorter peptides than was HLA-B*44:03. Collectively, these data highlight a mechanism through which polymorphism within an HLA class I supertype can diversify determinant selection and immune responses by varying peptide length preferences.
- Centre for Immunotherapy and Vaccine Development, Queensland Institute of Medical Research, Brisbane, Queensland 4029, Australia.
Organizational Affiliation: