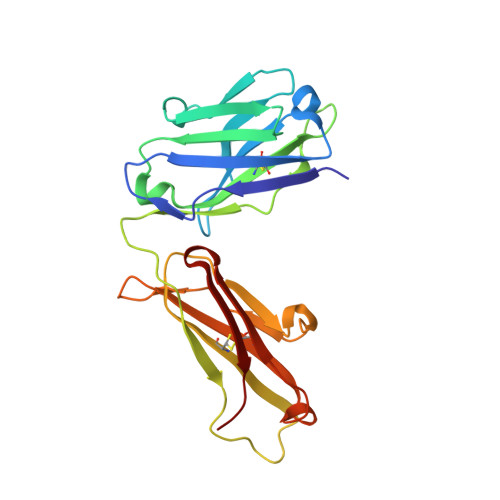
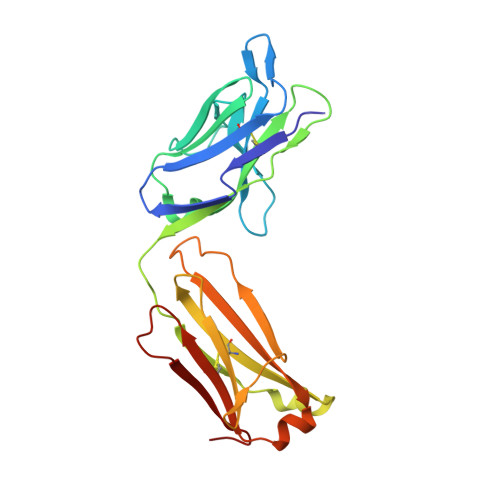
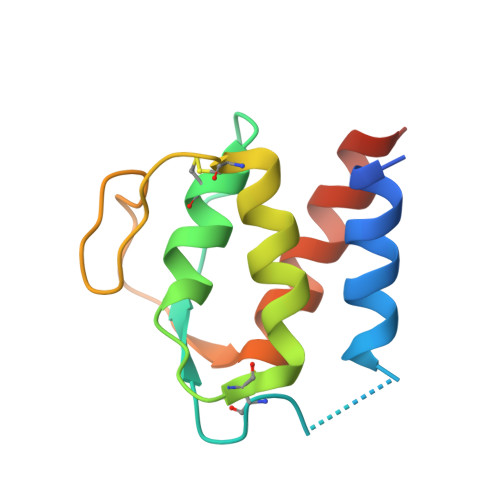
Structural Basis of Signaling Blockade by Anti-IL-13 Antibody Lebrikizumab.
Ultsch, M., Bevers, J., Nakamura, G., Vandlen, R., Kelley, R.F., Wu, L.C., Eigenbrot, C.(2013) J Mol Biology 425: 1330-1339
- PubMed: 23357170 Search on PubMed
- DOI: https://doi.org/10.1016/j.jmb.2013.01.024
- Primary Citation Related Structures:
4I77 - PubMed Abstract:
The cytokine interleukin 13 (IL-13) is a major effector molecule for T-helper type 2 inflammation and is pathogenic in allergic diseases such as asthma. The effects of IL-13 are mediated via a pathway that is initiated by binding to a heterodimeric receptor consisting of IL-13Rα1 and IL-4Rα. Antibodies raised against IL-13 can block its inflammatory effects by interfering with binding to either of the two receptor polypeptides. Lebrikizumab is a monoclonal anti-IL-13 antibody that has shown clinical benefit in a phase II study for the treatment of moderate-to-severe uncontrolled asthma. Here we report the molecular structure of IL-13 in complex with the Fab from lebrikizumab by X-ray crystallography at 1.9Å resolution. We show that lebrikizumab inhibits IL-13 signaling by binding to IL-13 with very high affinity and blocking IL-13 binding to IL-4Rα. In addition, we use site-directed mutations to identify the most important antibody contributors to binding. Our studies define key features of lebrikizumab binding and its mechanism of action that may contribute to its clinical effects.
- Department of Structural Biology, Genentech Inc., 1 DNA Way, South San Francisco, CA 94080, USA.
Organizational Affiliation: