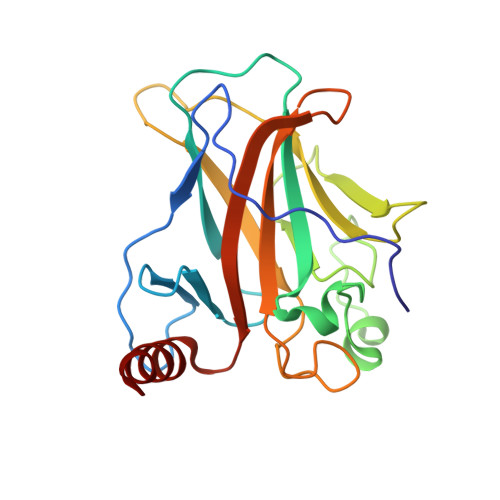
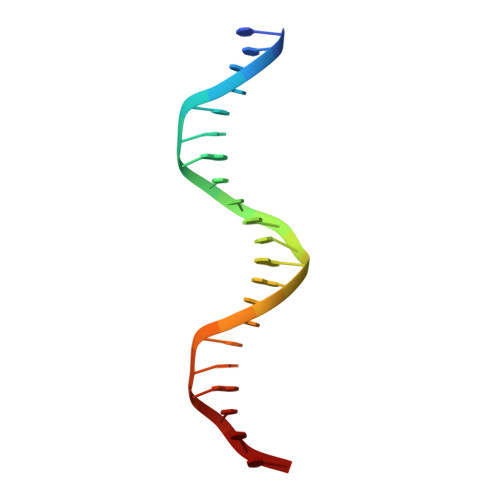
Structure of p53 binding to the BAX response element reveals DNA unwinding and compression to accommodate base-pair insertion.
Chen, Y., Zhang, X., Dantas Machado, A.C., Ding, Y., Chen, Z., Qin, P.Z., Rohs, R., Chen, L.(2013) Nucleic Acids Res 41: 8368-8376
- PubMed: 23836939 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1093/nar/gkt584
- Primary Citation Related Structures:
4HJE - PubMed Abstract:
The p53 core domain binds to response elements (REs) that contain two continuous half-sites as a cooperative tetramer, but how p53 recognizes discontinuous REs is not well understood. Here we describe the crystal structure of the p53 core domain bound to a naturally occurring RE located at the promoter of the Bcl-2-associated X protein (BAX) gene, which contains a one base-pair insertion between the two half-sites. Surprisingly, p53 forms a tetramer on the BAX-RE that is nearly identical to what has been reported on other REs with a 0-bp spacer. Each p53 dimer of the tetramer binds in register to a half-site and maintains the same protein-DNA interactions as previously observed, and the two dimers retain all the protein-protein contacts without undergoing rotation or translation. To accommodate the additional base pair, the DNA is deformed and partially disordered around the spacer region, resulting in an apparent unwinding and compression, such that the interactions between the dimers are maintained. Furthermore, DNA deformation within the p53-bound BAX-RE is confirmed in solution by site-directed spin labeling measurements. Our results provide a structural insight into the mechanism by which p53 binds to discontinuous sites with one base-pair spacer.
- Molecular and Computational Biology Program, Department of Biological Sciences, University of Southern California, Los Angeles, CA 90089, USA, Laboratory of Structural Biology, Key Laboratory of Cancer Proteomics of Chinese Ministry of Health, XiangYa Hospital & State Key Laboratory of Medical Genetics, Central South University, Changsha, Hunan 410008, China, Department of Chemistry, Norris Comprehensive Cancer Center, Department of Physics and Astronomy and Department of Computer Science, University of Southern California, Los Angeles, CA 90089, USA.
Organizational Affiliation: