Contribution of free energy of solvation to ligand affinity in new potent MMPs inhibitors.
Mori, M., Massaro, A., Calderone, V., Fragai, M., Luchinat, C., Mordini, A.To be published.
Experimental Data Snapshot
Starting Model: experimental
View more details
Macromolecule Content
Entity ID: 1 | |||||
|---|---|---|---|---|---|
| Molecule | Chains | Sequence Length | Organism | Details | Image |
| Macrophage metalloelastase | 158 | Homo sapiens | Mutation(s): 1 Gene Names: MMP12, HME EC: 3.4.24.65 | 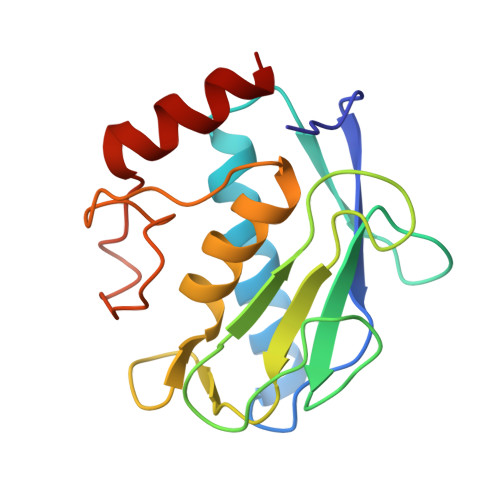 | |
UniProt & NIH Common Fund Data Resources | |||||
PHAROS: P39900 GTEx: ENSG00000262406 | |||||
Entity Groups | |||||
| Sequence Clusters | 30% Identity50% Identity70% Identity90% Identity95% Identity100% Identity | ||||
| UniProt Group | P39900 | ||||
Sequence AnnotationsExpand | |||||
Reference Sequence | |||||
| Ligands 3 Unique | |||||
|---|---|---|---|---|---|
| ID | Chains | Name / Formula / InChI Key | 2D Diagram | 3D Interactions | |
| KLJ Download:Ideal Coordinates CCD File | G [auth A] | N-hydroxy-N~2~-{[2-(4-methoxyphenyl)ethyl]sulfonyl}glycinamide C11 H16 N2 O5 S LMKSZZKXCRCXFM-UHFFFAOYSA-N |  | ||
| ZN Download:Ideal Coordinates CCD File | B [auth A], C [auth A] | ZINC ION Zn PTFCDOFLOPIGGS-UHFFFAOYSA-N |  | ||
| CA Download:Ideal Coordinates CCD File | D [auth A], E [auth A], F [auth A] | CALCIUM ION Ca BHPQYMZQTOCNFJ-UHFFFAOYSA-N |  | ||
| Length ( Å ) | Angle ( ˚ ) |
|---|---|
| a = 50.989 | α = 90 |
| b = 61.107 | β = 114.5 |
| c = 54.213 | γ = 90 |
| Software Name | Purpose |
|---|---|
| CrysalisPro | data collection |
| MOLREP | phasing |
| REFMAC | refinement |
| MOSFLM | data reduction |
| SCALA | data scaling |