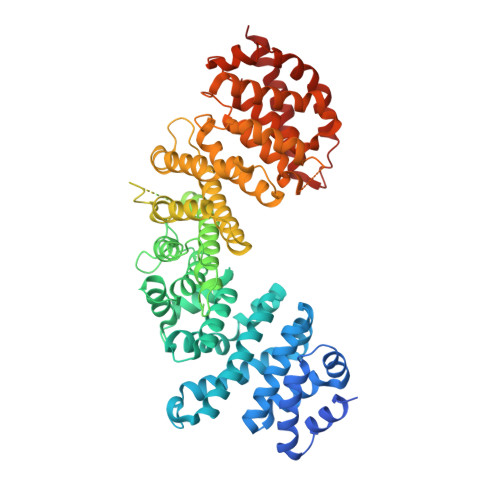Synchronizing nuclear import of ribosomal proteins with ribosome assembly.
Kressler, D., Bange, G., Ogawa, Y., Stjepanovic, G., Bradatsch, B., Pratte, D., Amlacher, S., Strauss, D., Yoneda, Y., Katahira, J., Sinning, I., Hurt, E.(2012) Science 338: 666-671
- PubMed: 23118189
- DOI: https://doi.org/10.1126/science.1226960
- Primary Citation Related Structures:
4GMN, 4GMO - PubMed Abstract:
Ribosomal proteins are synthesized in the cytoplasm, before nuclear import and assembly with ribosomal RNA (rRNA). Little is known about coordination of nucleocytoplasmic transport with ribosome assembly. Here, we identify a transport adaptor, symportin 1 (Syo1), that facilitates synchronized coimport of the two 5S-rRNA binding proteins Rpl5 and Rpl11. In vitro studies revealed that Syo1 concomitantly binds Rpl5-Rpl11 and furthermore recruits the import receptor Kap104. The Syo1-Rpl5-Rpl11 import complex is released from Kap104 by RanGTP and can be directly transferred onto the 5S rRNA. Syo1 can shuttle back to the cytoplasm by interaction with phenylalanine-glycine nucleoporins. X-ray crystallography uncovered how the α-solenoid symportin accommodates the Rpl5 amino terminus, normally bound to 5S rRNA, in an extended groove. Symportin-mediated coimport of Rpl5-Rpl11 could ensure coordinated and stoichiometric incorporation of these proteins into pre-60S ribosomes.
- Biochemie-Zentrum der Universität Heidelberg, Im Neuenheimer Feld 328, Heidelberg D-69120, Germany. dieter.kressler@unifr.ch
Organizational Affiliation: