Biochemical and Structural Insights into Xylan Utilization by the Thermophilic Bacterium Caldanaerobius polysaccharolyticus.
Han, Y., Agarwal, V., Dodd, D., Kim, J., Bae, B., Mackie, R.I., Nair, S.K., Cann, I.K.(2012) J Biological Chem 287: 34946-34960
- PubMed: 22918832 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.M112.391532
- Primary Citation Related Structures:
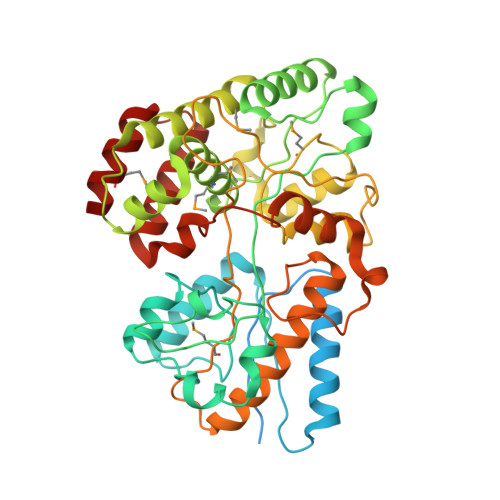
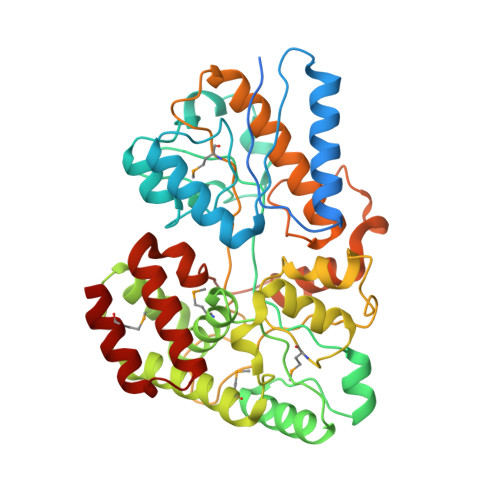
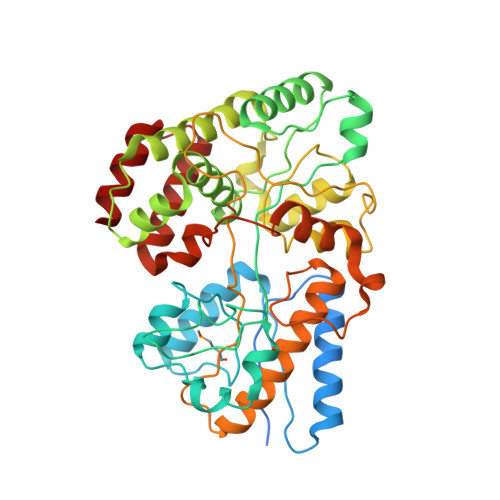
4G68 - PubMed Abstract:
Hemicellulose is the next most abundant plant cell wall component after cellulose. The abundance of hemicellulose such as xylan suggests that their hydrolysis and conversion to biofuels can improve the economics of bioenergy production. In an effort to understand xylan hydrolysis at high temperatures, we sequenced the genome of the thermophilic bacterium Caldanaerobius polysaccharolyticus. Analysis of the partial genome sequence revealed a gene cluster that contained both hydrolytic enzymes and also enzymes key to the pentose-phosphate pathway. The hydrolytic enzymes in the gene cluster were demonstrated to convert products from a large endoxylanase (Xyn10A) predicted to anchor to the surface of the bacterium. We further use structural and calorimetric studies to demonstrate that the end products of Xyn10A hydrolysis of xylan are recognized and bound by XBP1, a putative solute-binding protein, likely for transport into the cell. The XBP1 protein showed preference for xylo-oligosaccharides as follows: xylotriose > xylobiose > xylotetraose. To elucidate the structural basis for the oligosaccharide preference, we solved the co-crystal structure of XBP1 complexed with xylotriose to a 1.8-Å resolution. Analysis of the biochemical data in the context of the co-crystal structure reveals the molecular underpinnings of oligosaccharide length specificity.
- Energy Biosciences Institute, University of Illinois, Urbana, Illinois 61801; Institute for Genomic Biology, University of Illinois, Urbana, Illinois 61801.
Organizational Affiliation: