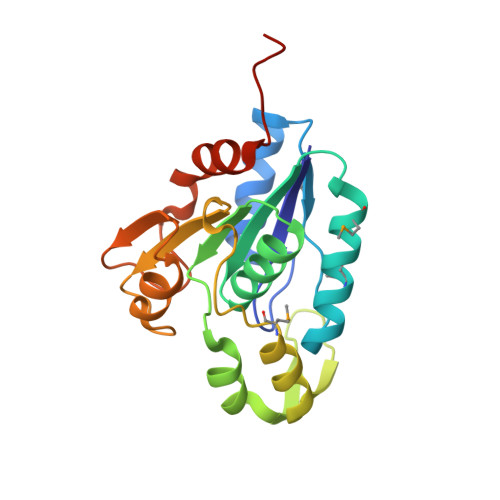
Crystal structure of the esterase YqiA (YE3661) from Yersinia enterocolitica, Northeast Structural Genomics Consortium Target YeR85 (CASP Target)
Forouhar, F., Lew, S., Seetharaman, J., Shastry, R., Kohan, E., Maglaqui, M., Mao, L., Xiao, R., Acton, T.B., Everett, J.K., Montelione, G.T., Tong, L., Hunt, J.F., Northeast Structural Genomics Consortium (NESG)To be published.