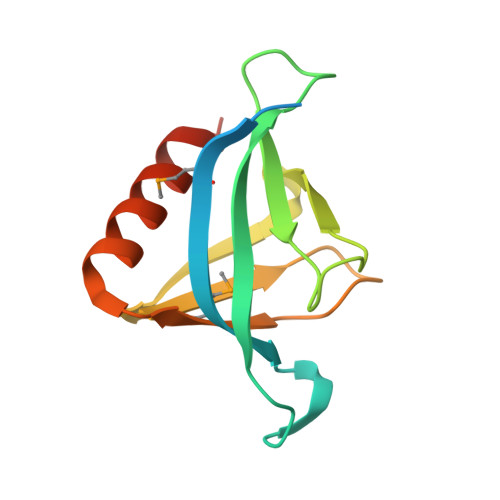
Structural Analysis of SHARPIN, a Subunit of a Large Multi-protein E3 Ubiquitin Ligase, Reveals a Novel Dimerization Function for the Pleckstrin Homology Superfold.
Stieglitz, B., Haire, L.F., Dikic, I., Rittinger, K.(2012) J Biological Chem 287: 20823-20829
- PubMed: 22549881 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1074/jbc.M112.359547
- Primary Citation Related Structures:
4EMO - PubMed Abstract:
SHARPIN (SHANK-associated RH domain interacting protein) is part of a large multi-protein E3 ubiquitin ligase complex called LUBAC (linear ubiquitin chain assembly complex), which catalyzes the formation of linear ubiquitin chains and regulates immune and apoptopic signaling pathways. The C-terminal half of SHARPIN contains ubiquitin-like domain and Npl4-zinc finger domains that mediate the interaction with the LUBAC subunit HOIP and ubiquitin, respectively. In contrast, the N-terminal region does not show any homology with known protein interaction domains but has been suggested to be responsible for self-association of SHARPIN, presumably via a coiled-coil region. We have determined the crystal structure of the N-terminal portion of SHARPIN, which adopts the highly conserved pleckstrin homology superfold that is often used as a scaffold to create protein interaction modules. We show that in SHARPIN, this domain does not appear to be used as a ligand recognition domain because it lacks many of the surface properties that are present in other pleckstrin homology fold-based interaction modules. Instead, it acts as a dimerization module extending the functional applications of this superfold.
- Division of Molecular Structure, Medical Research Council National Institute for Medical Research, The Ridgeway, London NW7 1AA, United Kingdom.
Organizational Affiliation: