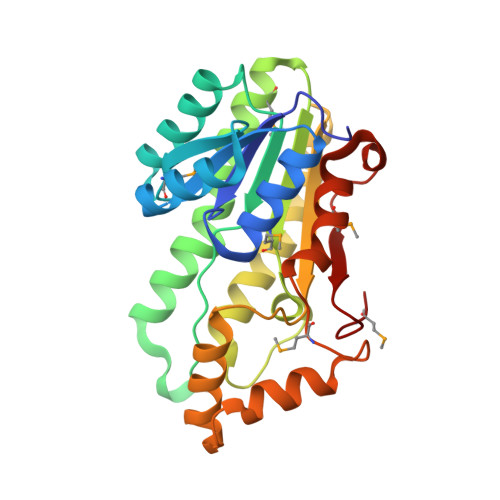
Crystal structure of a probable sorbitol dehydrogenase from Sinorhizobium meliloti 1021
Kumar, P.R., Banu, N., Bhosle, R., Bonanno, J., Chamala, S., Chowdhury, S., Gizzi, A., Glen, S., Hammonds, J., Hillerich, B., Love, J.D., Matikainen, B., Seidel, R., Toro, R., Zencheck, W., Almo, S.C.To be published.