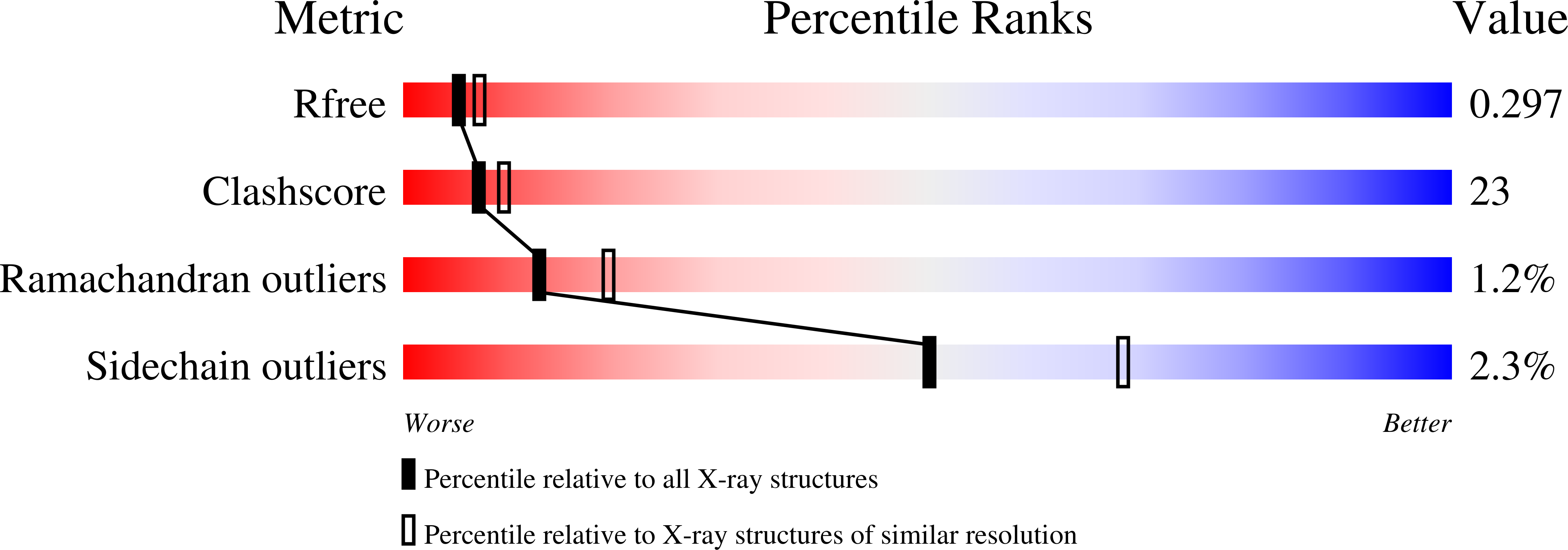
A strategy based on nucleotide specificity leads to a subfamily-selective and cell-active inhibitor ofN6-methyladenosine demethylase FTO.
Toh, J.D.W., Sun, L., Lau, L.Z.M., Tan, J., Low, J.J.A., Tang, C.W.Q., Cheong, E.J.Y., Tan, M.J.H., Chen, Y., Hong, W., Gao, Y.G., Woon, E.C.Y.(2015) Chem Sci 6: 112-122
- PubMed: 28553460 Search on PubMedSearch on PubMed Central
- DOI: https://doi.org/10.1039/c4sc02554g
- Primary Citation Related Structures:
4CXW, 4CXX, 4CXY - PubMed Abstract:
The AlkB family of nucleic acid demethylases are of intense biological and medical interest because of their roles in nucleic acid repair and epigenetic modification. However their functional and molecular mechanisms are unclear, hence, there is strong interest in developing selective inhibitors for them. Here we report the identification of key residues within the nucleotide-binding sites of the AlkB subfamilies that likely determine their substrate specificity. We further provide proof of principle that a strategy exploiting these inherent structural differences can enable selective and potent inhibition of the AlkB subfamilies. This is demonstrated by the first report of a subfamily-selective and cell-active FTO inhibitor 12 . The distinct selectivity of 12 for FTO against other AlkB subfamilies and 2OG oxygenases shall be of considerable interest with regards to its potential use as a functional probe. The strategy outlined here is likely applicable to other AlkB subfamilies, and, more widely, to other 2OG oxygenases.
- Department of Pharmacy , National University of Singapore , 18 Science Drive 4 , 117543 , Singapore . Email: esther.woon@nus.edu.sg ; ; Tel: +65 6516 2932.
Organizational Affiliation: